Tactics of Managing Postinfarction Interventricular Septal Rupture
The aim of the study was to develop tactics of surgical treatment of postinfarction interventricular septal (IVS) ruptures.
Materials and Methods. Eighteen patients with the diagnosis of “postinfarction interventricular septal rupture” have been examined and operated on. The majority of them were men, 14 in number (77.7%), and only 4 women (22.3%). The average age was 55.6±6.4 years. The time of hospitalization from the IVS rupture onset varied from 4 days to 3.5 months. Clinically, there was noted left ventricular (4 cases) or biventricular (14 cases) failure of different degree with edema of lower extremities and/or uni- or bilateral hydrothorax. Patients were performed intra-aortic balloon counterpulsation in order to stabilize clinical state and support hemodynamics.
Results. The assessment of the initial clinical state of the patients with postinfarction rupture of the IVS showed an extremely severe course of ischemic heart disease. Symptoms of severe heart failure (III–IV FC according to NYHA) were noted in all patients. Besides, 14 patients had angina pectoris (III–IV FC according to CCS) being evidence of a heavy damage of the coronary arterial bed. The expected mortality risk for our patients was 12.43±3.0% according to the EuroSCORE. Coronography demonstrated that the anterior descending artery (100%) and right coronary artery (62%) were the most commonly injured territories.
Our own experience and the data obtained from the literature enabled us to develop the tactics of surgical treatment of these patients.
Introduction
Surgical treatment of postinfarction interventricular septal (IVS) rupture is one of the most complicated and unsolved problems in the current cardiosurgery. The rupture manifested itself by the acute emergence of blood flow to the right ventricle (RV) from the left ventricle (LV) (left-to-right shunting) resulting in rapidly progressive heart failure [1].
Despite the improvement of the methods for early diagnosis and treatment of acute myocardial infarction (MI) the occurrence rate of this complication remains high (1–3%) [2, 3]. At the same time, the chance of favorable outcome in patients treated for IVS rupture is not great and without timely rendered medical aid the rupture leads to inevitable death.
Surgical treatment of patients with this pathology is performed by life-saving indications and is accompanied by high lethality rate of 25–70% [3, 4]. In this connection, this problem continues to be a surgical challenge demanding adequate solution.
Many investigators [1, 2, 5] propose to perform operative intervention by urgent indications as soon as the diagnosis of postinfarction IVS rupture is established regardless of the heart failure intensity. But it is known that the operation done within the first week of this condition has a 75% lethality rate whereas its performance 3 weeks later reduces this figure to 16% [1, 2, 6].
The task of the operative treatment of postinfarction IVS rupture is not only to save the patient’s life but to rehabilitate him, i.e. to maintain or restore his ability to work. And, as the experience of some researchers shows [7], it is possible if the operation is done at the early stages of the rupture before the development of severe polyorganic insufficiency.
At present, the following problems of surgical management are left unresolved: adequate assessment of the severity of the initial patients’ state before the operation, tactics of managing patients with postinfarction IVS rupture in the preoperative and postoperative periods, the possibility and necessity of applying inter-aortal balloon counterpulsation, the choice of optimal terms of reconstructive operative intervention to eliminate the defect. Still pending are a number of technical aspects of operative treatment associated with the necessity of myocardial revascularization, radical correction of postinfarction IVS rupture, and correction of the concomitant valvular pathology [1, 6, 8].
The aim of the study was to summarize the accumulated experience and to define the tactics of surgical management of postinfarction interventricular septal ruptures.
Materials and Methods
Since 1989, 18 patients with the diagnosis of “postinfarction interventricular septal rupture” were operated on at the Specialized Cardiosurgical Clinical Hospital (Nizhny Novgorod). The majority of patients were men, 14 in number (77.7%), and only 4 women (22.3%). The average age was 55.6±6.4 years. Eleven patients suffered anterior MI, 6 posterior MI, and 1 had extensive anteroposterior MI. Later, formation of LV aneurysm was revealed by echoCG in 7 patients.
Rupture of the IVS occurred within the first week of acute MI in 10 patients, in the second week in 6, and in the third week in 2. The time of hospitalization to the cardiosurgical department since the onset of the IVS rupture varied from 4 days to 3.5 months. Clinically, left ventricular (4 cases) or bilateral (14 cases) insufficiency of different degree with edema of the lower extremities and/or one- or both-sided hydrothorax were noted. In order to stabilize the clinical state and support hemodynamics, patients underwent intra-aortic balloon counterpulsation. Determination of the exact IVS rupture localization, evaluation of the patients’ clinical state, function of the atrioventricular valves, LV geometry, the degree of the coronary artery bed damage were performed using invasive and noninvasive investigation techniques.
All patients presented complaints of the marked feeling of getting not enough air, interruptions in the work of the heart, enhanced heartbeat. A harsh systolic murmur was heard over the entire heart region with the epicenter in the 3d–4th intercostal space to the left from the sternum or in the apex area.
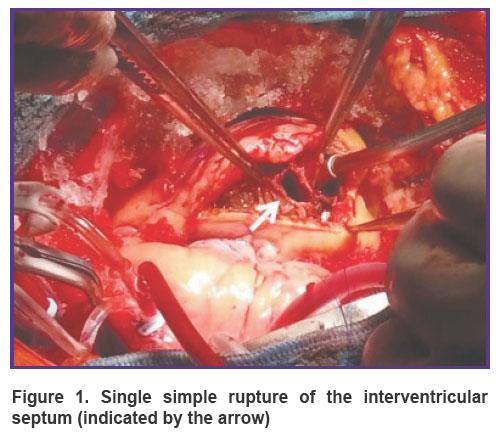
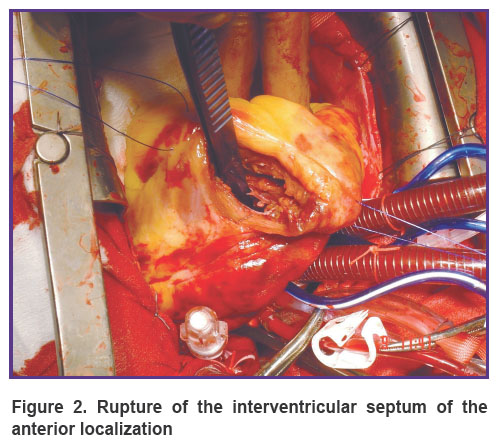
Single simple IVS ruptures were detected in 10 patients (Figure 1). The rupture size was from 1.5 to 2.5 cm. Single complex (i.e. having a convoluted flow and IVS delamination) ruptures were found in 8 patients. The rupture diameter ranged from 2.0 to 4.0 cm. Multiple ruptures have not been noted. IVS ruptures of anterior localization (high, medium, central outflow trabecular, inferior apical trabecular) were observed in 12 patients (Figure 2), ruptures of the posterior localization (sine, sine-trabecular, trabecular, tunnel-shaped) were found in 6 persons.
|
Figure 1. Single simple rupture of the interventricular septum (indicated by the arrow) |
|
Figure 2. Rupture of the interventricular septum of the anterior localization |
The study complies with the Declaration of Helsinki and was performed following the approval of the Ethics Committee of the Specialized Cardiosurgical Clinical Hospital. Written informed consent was obtained from every patient.
All patients with the postinfarction rupture of the IVS were performed transthoracic and transesophageal echoscopic cardiography (echoCG), pressure was measured in the pulmonary artery (PAP) intraoperatively before and after plastic repair of the IVS rupture. In such patients, echoCG examination shows moderate dilatation of the right and left parts of the heart, hypertrophy of the LF myocardium, signs of hypokinesia and akinesia of the anteroapical and lateral LV walls in patients with anterior heart aneurysms, and the signs of akinesia of the posterolateral parts if there is aneurysm of the LV posteroinferior wall.
The following averaged findings were obtained on echoCG examination before the operation: end-diastolic volume (EDV) — 134.2±69.8 ml, ejection fraction (EF) — 44.6±8.43%, PAP — 47.6±3.6 mm Hg.
A total of 18 operations were performed for postinfarction IVS rupture. All operations were done under the conditions of artificial circulation, normothermia, pharmaco-hypothermic cardioplegia. Mean duration of perfusion was 121.6±20.4 min; aortic clamping — 91.0±10.05 min.
The operation for elimination of postinfarction IVS rupture was performed on 7 patients within 3 weeks after MI (10.0±7.0 days on average), the rest 11 patients were operated on 4–8 weeks after the occurrence of the postinfarction rupture.
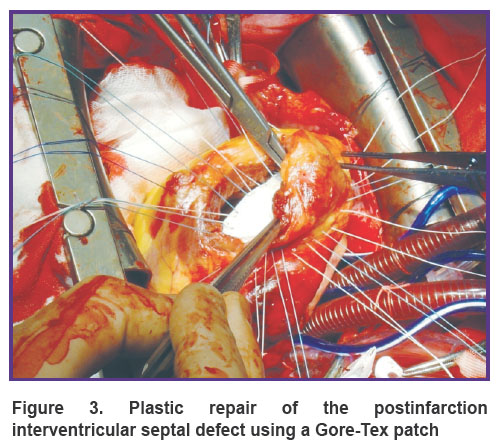
Dura mater was used for plastic repair of the rupture in 4 operative interventions, xenopericardium in 3 operations, and in the rest 11 cases Gore-Tex patches were applied (Figure 3). Surgical approach to the rupture was through the right parts of the heart in 7 cases and through the LV in 11 cases.
|
Figure 3. Plastic repair of the postinfarction interventricular septal defectusing a Gore-Tex patch |
Coronary bypass of 1–3 arteries was performed in 17 patients.
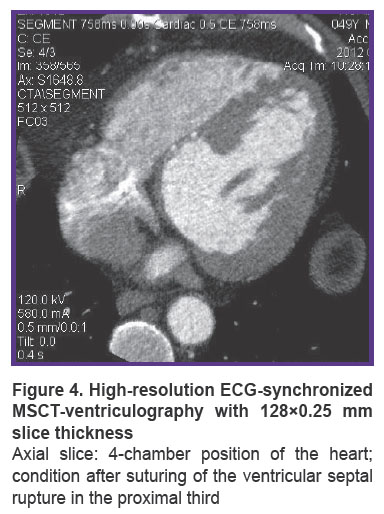
In all operations, the IVS rupture was sutured with U-type Teflon-pledgeted sutures. In 4 patients suturing was supplemented with IVS plastic repair with a patch located on the LV side (Figure 4).
Resection of postinfarction LV aneurysm with the following plastic repair of the rupture was performed in 9 patients. In one case, plastic repair was done without coronary bypass. In 4 cases, Dor’s repair was performed with implantation of the second Gore-Tex patch; in 4 cases the repair was done according to Jatene–Cooley (internal purse-string suture + two-layer twisted suture of the RV wall); in 1 case, aneurysm and delamination of the external LV wall were sutured with pledgets and twisted sutures.
Results and Discussion
After the operation for postinfarction IVS rupture, 4 patients died within the hospitalization period. Lethality was 22.2%. Three patients died on days 2, 3, and 7 after the operation from progressive heart failure. In two of them the condition became worse due to recanalization of the IVS rupture. The fourth died on day 11 due to progressive polyorganic insufficiency after the adequately performed operation because of untimely diagnosed complexity of the rupture having a high anterior localization and late admittance for surgical treatment (3.5 months after the septal rupture). All survived patients demonstrated improvement of the main hemodynamic parameters with normalization and reduction of pressure in the pulmonary artery.
Assessment of the initial clinical state of the patients with postinfarction IVS rupture demonstrated an extremely heavy course of IHD in this group of patients. Symptoms of severe heart failure (III–IV class according to NYHA) were observed in all patients. Besides, 14 patients had angina pectoris (III–IV class according to CCS) being evidence of a heavy damage of the coronary arterial bed. The expected mortality risk for these patients was 12.43±3.0% according to the EuroSCORE scale. Coronography demonstrated that the anterior descending artery (100%) and right coronary artery (62%) were the most commonly injured territories explaining the reason of anterior and posterior MI with the subsequent development of IVS rupture (Figure 5).
|
Figure 5. Coronarography data of the patients with postinfarction interventricular septal rupture: occlusion of the anterior descending artery |
After the operative intervention, the echoCG indices were as follows: EDV — 121.5±18.34 ml; EF — 49.0±7.2%; PAP — 36.5±16.37 mm Hg. Average hospital stay was 45.7±21.5 days.
The main complications after the operation were: acute cardiac insufficiency demanding intra-aortic balloon counterpulsation and inotropic therapy in 8 cases; recanalization of the IVS rupture in 4 cases; signs of polyorganic insufficiency in 3 cases; postoperative wound suppuration in 2 cases; pleurisy in 2 cases; pneumothorax and atrial fibrillation in 1 case.
The obtained results and the observed complications depend to a great extent on the surgical tactics applied. It is a cornerstone of managing patients with postinfarction IVS rupture and is based on the solution of the three important tasks: optimal terms of performing the operation after the rupture onset, strict observation of the technical aspects of the reconstructive operation; evaluation of the completeness of the reconstruction of the damaged cardiac structures [9].
As a rule, an IVS rupture is a consequence of septum necrotizing throughout the entire thickness. The time and localization depend directly on the trophic disturbances of the definite myocardial area. Occlusion of the anterior descending and right coronary arteries is most commonly associated with the IVS rupture: 42 and 46% cases, respectively [6].
In our work, the choice of the time for surgical correction of IVS rupture was determined by the severity of hemodynamic disturbances. In case of progressive heart failure, emergency surgical intervention was performed. Emergency operation was indicated to 11 patients. It was performed on days 4–14 after the IVS rupture. Four patients died. Wait-and-see attitude was used in relation to the rest 7 patients as they demonstrated stable hemodynamics. These patients underwent operative intervention 4–16 weeks after the IVS rupture.
Two main types of IVS ruptures are usually distinguished: simple and complex. In a simple rupture, the canal of communication opens at every systolic contraction, gross hemorrhages or lacerations are absent, as a rule, and the outflow holes in RV and LV are located on the same level. Complex ruptures occur when interventricular communication has irregular curved flow with the canal which can extend to the regions distant from the initial area of acute MI. Complex ruptures were noted to complicate diaphragmatic infarctions more often, whereas simple ruptures appear usually after MI involving the anterior wall.
The choice of plastic repair method depends on the IVS rupture localization determining also the character of the approach to the rupture. To correct anterior rupture we used left ventriculotomy through the infarcted myocardium (at different times) in 9 patients or through the scar field (at later periods of time) in 4 cases. This approach is considered optimal for adequate visualization of the heart cavity. When the rupture in 4 patients was of posterior localization, access to the defect was achieved by a longitudinal incision through posterobasal aneurysm (1 patient) or through the infarction zone (3 patients). In 1 case the access to the defect was through the right ventricle.
The first two plastic corrections (3 operations) of postinfarction IVS rupture were done using a dura mater patch, the rest (15 operations) with a Gore-Tex patch or from xenopericardium using Teflon-pledgeted U-sutures. The front row of the sutures was passed behind the IVS to the right ventricle. Concurrently with the IVS rupture plasty, aneurysmectomy and LV reconstruction were conducted in 8 patients: according to Dor in 4 patients with the anterior localization of aneurysm, according to Jatene–Cooley in 3 patients (1 with anterior and 2 with posterior localization), and in 1 case, suturing of aneurysm and delamination of the external LV wall were performed using pledgets and twisted sutures. In 17 cases, revascularization of the myocardium was fulfilled.
Undoubtedly, patients with postinfarction IVS rupture are referred to the cohort of the sick people with the most severe IHD and extremely poor prognosis if the disease runs its natural course. It is worth mentioning that in patients with diffused damage of the coronary arteries, IVS ruptures occur less frequently than in those with a single damaged vessel but without the developed collateral circulation [5, 6]. IVS rupture results in the left-to-right shunting. As a result, the LV which may be involved in acute MI bears additional overloads. The rupture size influences directly the volume of the discharged blood and hemodynamic consequences. The volume of the shunted blood will also depend on the resistance in the pulmonary vessels and systolic arterial pressure in the systemic circulation.
A typical clinical picture of IVS rupture includes a sudden worsening of the state in persons suffered MI, increasing breathlessness, sometimes pain in the chest, frequently with the development of cardiogenic shock in presence of pansystolic murmur which is best heard along the left parasternal line [4, 10]. Elderly age, stroke in the past history, persisting ST-segment elevation and an increased level of cardiac markers, tachycardia, hypotension and a high Killip class of acute cardiac insufficiency, absence of complete revascularization during hospital stay are referred to unfavorable prognostic markers [2, 8, 11].
Surgical treatment of IVS rupture is conducted according to life-saving indications, operative lethality is 25–70%. The time of surgical correction of IVS rupture is determined by the degree of hemodynamic disturbances (progressive cardiac, polyorganic insufficiency). In spite of the fact that the American Association for Cardiovascular Surgery recommended in 2013 to perform emergency surgery irrespective of the hemodynamic conditions, this question has been under discussion till present time. Thus, lethality in surgical correction of postinfarction IVS rupture during 1 week of MI reaches 75%, 3 weeks later it reduces to 16% [5, 10].
Conclusion
Early surgical treatment of postinfarction interventricular septal rupture is conducted according to life-saving indications to the patients with high pulmonary pressure and if cardiac and polyorganic insufficiency grows progressively worse.
The main purpose of surgical treatment is reliably close (close to) the septal rupture.
It is reasonable to use differential approach to the choice of the time of operation performance: actively (urgently) if the events of cardiac failure are growing worse, or using a wait-and-see approach if hemodynamics and clinical state of the patient are stable.
The efficacy of surgical treatment depends on the completeness of the entire complex of morphological changes in the heart.
Study funding. The work was not supported by any financial source.
Conflicts of interest. The authors have no conflicts of interest to declare.
References
- Zhbanov I.V., Molochkov A.V., Aksel’rod B.A., Shmyrin M.M., Fedulova S.V., Shabalkin B.V. Surgical treatment of postinfarctial ruptures of interventricular septum. Khirurgiya. Zhurnal imeni N.I. Pirogova 2013; 2: 35–39.
- Goyal A., Menon V. Contemporary management of post-MI ventricular septal rupture. J Am Coll Cardiol 2018.
- Moreyra A.E., Huang M.S., Wilson A.C., Deng Y., Cosgrove N.M., Kostis J.B. Trends in incidence and mortality rates of ventricular septal rupture during acute myocardial infarction. Am J Cardiol 2010; 106(8): 1095–1100, https://doi.org/10.1016/j.amjcard.2010.06.013.
- Honda S., Asaumi Y., Yamane T., Nagai T., Miyagi T., Noguchi T., Anzai T., Goto Y., Ishihara M., Nishimura K., Ogawa H., Ishibashi-Ueda H., Yasuda S. Trends in the clinical and pathological characteristics of cardiac rupture in patients with acute myocardial infarction over 35 years. J Am Heart Assoc 2014; 3(5): e000984, https://doi.org/10.1161/jaha.114.000984.
- Lundblad R., Abdelnoor M., Geiran O.R., Svennevig J.L. Surgical repair of postinfarction ventricular septal rupture: risk factors of early and late death. J Thorac Cardiovasc Surg 2009; 137(4): 862–868, https://doi.org/10.1016/j.jtcvs.2008.09.008.
- Alshibaya M.M., Kovalenko O.A., Vishchipanov S.A. Surgical treatment of complicated forms of postinfarction heart aneurysm. Byulleten’ NTsSSKh im. A.N. Bakuleva RAMN 2009; 10(S6): 64.
- Noguchi K., Yamaguchi A., Naito K., Yuri K., Adachi H. Short-term and long-term outcomes of postinfarction ventricular septal perforation. Gen Thorac Cardiovasc Surg 2012; 60(5): 261–267, https://doi.org/10.1007/s11748-011-0882-1.
- Rabinovich R.M., Alshibaya M.M. Heart rupture: a case of successful medical treatment of acute rupture of left ventricular free wall during acute myocardial infarction with following surgical correction. Kreativnaya kardiologiya 2015; 3: 67–79.
- Alshibaya M.M., Krimov K.V., Musin D.E., Cheishvili Z.M. Modified method of surgical correction of interventricular septum postinfarction. Annaly hirurgii 2008; 2: 78–80.
- Sajja L.R., Mannam G.C., Gutti R.S., Goli N.R., Sompalli S., Penumatsa R.R. Postinfarction ventricular septal defect: patch repair with infarct exclusion. Asian Cardiovasc Thorac Ann 2008; 16(3): 215–220, https://doi.org/10.1016/s0022-5223(95)70054-4.
- Rohn V., Spacek M., Belohlavek J., Tosovsky J. Cardiogenic shock in patient with posterior postinfarction septal rupture-successful treatment with extracorporeal membrane oxygenation (ECMO) as a ventricular assist device. J Card Surg 2009; 24(4): 435–436, https://doi.org/10.1111/j.1540-8191.2008.00710.x.