Inflammatory Complications of the Abdominal Wall Prosthetic Repair: Diagnostics, Treatment, and Prevention (Review)
A number of issues in diagnosis, treatment, and prevention of purulent and septic complications in implantation of mesh are currently very far from being resolved. Patients with acute or chronic inflammatory processes associated with a mesh are of special concern, as this situation can completely discredit the possibilities of the abdominal wall reconstruction and the use of synthetic materials for this purpose, casting doubt on the accumulated knowledge, experience of the clinic and the surgeons’ qualification.
Hereinafter the most up-to-date and clarified classifications of inflammatory complications are demonstrated. A systemic approach to the problem of clear stratification of patients depending on the risk of complication is provided. Differences and controversial opinions regarding terminology, latest concepts and definitions in the surgery of abdominal wall hernias are presented.
Possibilities of anticipating potential complications are specified. Identification of a special group of patients with large and complex, multiple defects of the abdominal wall is justified. The most effective methods of diagnosing the complications and the results of their application are assessed.
The definitions of an acute inflammatory process associated with endoprosthesis implantation and of a chronic paraprosthetic infection are given. The modern approaches to treatment including generally accepted methods and controversial opinions are described. Options of mesh excision, repeated prosthetic repair of the abdominal wall are presented; successes and failures depending on the chosen strategy are shown. The strategy of endoprosthesis saving is grounded; the underlying methods (NPWT — negative pressure wound therapy), their benefits and drawbacks are described. The most important areas of prevention of purulent and septic complications in herniology are specified.
Technical issues including the comparison of surgical methods, a certain range of techniques that can objectively eliminate the existing risks are provided. A specificity of endoprosthesis choice depending on the accumulated findings about mesh material, its manufacturing methods and properties is emphasized. The concept of implant prophylaxis is disclosed.
Introduction
Incisional hernias are a frequent outcome of abdominal surgery. Up to 20% of laparotomies result in hernias and among high-risk groups of patients the frequency of incisional hernia exceeds 35% [1]. Annually 4–5 millions of laparotomies are performed in the USA and they account for about 500 thousand incisional hernias [1, 2]. Their frequency in different reviews ranges from 3 to 50% [2]. The traditional surgical methods based on closing the defect with the patient’s own tissues have low effectiveness resulting in up to 63% of recurrent hernias [2]. The main method of treating these patients was prosthetic repair that could reduce this indicator for a three-year period of observation by 1 to 24% depending on a specific category of patients and the applied method of surgery [2]. Possibilities of prophylactic mesh implantation to prevent formation of hernias are being actively studied [3]. The abdominal wall reconstruction is a method of choice in modern herniology which has been proved by evidence-based medicine [4].
Unfortunately, some patients having undergone implantation of mesh have complications. A special category of complications is purulent and septic complications that have the biggest impact on clinical prediction, life quality parameters, long-term surgery outcomes, recurrent hernias and the need for repeated surgery. Patients with large hernias, chronic infection and intestinal fistulas are a group of special concern. According to the data of meta-analysis [5], a complex abdominal wall reconstruction under conditions of the surgical field contamination results in 46% of wound complications.
The flora is usually represented by Staphylococcus aureus (including MRSA — methicillin-resistant Staphylococcus aureus), Staphylococcus coagulase negative, Enterococcus (VRE — vancomycin-resistant Enterococci) [6–12], Pseudomonas aeruginosa [13], Proteus, Klebsiella [7, 12], Streptococcus beta-hemolytic, Corynebacterium, Acinetobacter baumannii, Escherichia coli [7, 11], in exceptional cases — Clostridium perfringens [12] or fungi Candida [6, 14]. Development of paraprosthetic infection includes the stages of mesh colonization and biofilm formation [15, 16]. It is a biofilm variant of infectious process development that underlies the difficulties and failures in its treatment [6]. The possibilities of antibacterial therapy are seriously limited by low penetration of medications into the biofilm. Furthermore, minimal suppressing concentrations of medications for biofilm forms of microorganisms exceed the ones for planktonic forms in ten and hundred times [6].
Errors and complications in treating patients with the abdominal wall hernias should be given special attention [17, 18]. According to the apt words of the leading experts, infections in the surgical site are Achilles’ heel of absolutely all operations targeted at the abdominal wall reconstruction in regard to hernias [19]. A number of issues in diagnosis, treatment, and prevention of purulent and septic complications in implantation of mesh are currently very far from being resolved. [20]. It is well-known that frequency of events in the surgical site depends on the chosen surgical technique, although the authors give different results [21]. The category of patients of highest concern is represented by the patients with large hernias and acute or chronic inflammatory process associated with a previously implanted mesh [22].
Surgical treatment of patients with rather large hernia defects requires using specific and non-standard technical solutions [23]. Such complicated operations naturally entail a number of problems. On the other hand, inflammatory complications can also develop after typical variants of prosthetic repair. The above-mentioned clinical picture can appear at the most inappropriate period of treatment [24]. Negative clinical results [25] in certain situation can completely discredit the possibilities of the abdominal wall reconstruction and the use of synthetic materials, question the accumulated knowledge, experience of the clinic and the surgeons’ qualification [26–28].
In a number of cases unpleasant consequences can be observed in the long-term perspective after the operation, in 2–12 years after the operation on hernia [12, 29, 30]. For instance, mesh migration from the implantation region to the abdominal cavity — the complication which is considered to be a severe one [31], can be accompanied with the destruction of the hollow organ wall [32–34]. In other cases, a purulent and septic process developed rather actively and early [35, 36]. Patients with large and complicated incisional hernias require special attention [22]. According to the data of meta-analyses, up to 11% of patients have the abdominal wall defects of more than 15 cm long [37]. The use of prosthetic repair in this category of patients is the only effective solution. Frequency of complications in this category ranges from 4 to 100% with the median of 32% [37]. Patients with hernias and obesity of degree III and IV have postoperative wound complications in 14–36% of cases, cardiovascular events in 44–72.6% and an abdominal compartment syndrome in 8.3–30.6% [38]. In some cases surgeons have to refuse from implantation of mesh because of a mesh-associated infection risk [39]. The issue of developing a strategy of prevention and treatment of purulent and septic complications associated with the abdominal wall prosthetic repair is one of the topical ones in abdominal surgery and herniology [40].
Classification of complications
There is no unified and generally accepted classification of complications of prosthetic repair today. The most relevant and essential current classifications are the ones by Clavien–Dindo and Accordion [41].
According to Clavien–Dindo Classification, complications are divided into 5 classes:
I — deviations from the normal course of postoperative period that do not require special therapy or intervention, including wound infection treatment;
II — problems requiring infusion/transfusion therapy, enteral and/or parenteral feeding;
III — situations that require surgical intervention (IIIa — under local anesthesia, IIIb — under general anesthesia);
IVa — organ dysfunction;
IVb — multiple organ failure;
V — lethal outcome.
Accordion’s classification subdivides the problems of postoperative period into two categories [41]. The first one (minor complications) includes classes I–III:
I — minimal invasive procedures and treatment of a wound infection;
II — complications that require pharmacotherapy;
III — endoscopic and/or intervention procedures without general anesthesia.
The second category (major complications) includes classes IV–VI:
IV — surgical intervention under general anesthesia with artificial lung ventilation and a single organ dysfunction;
V — surgical intervention under general anesthesia with artificial lung ventilation and a single organ dysfunction or multiple organ failure;
VI — lethal outcome.
Fluid collection associated with surgery on the abdominal wall (seroma) is considered separately and several types can be identified [42]. They are understood differently in different literature sources. Russian authors define them as any fluid collection in subcutaneous tissue which is identified either clinically or with ultrasonography; they are referred to complications [42]. This approach is justified, and small seromas without clinical manifestations correspond to Clavien–Dindo I, but are not considered by Accordion. Belokonev [43] defines seroma as a limited fluid collection after any surgery with wide mobilization of a cutaneous-subcutaneous flap. However, it should be noted that localization of seromas in the subcutaneous tissue is not the only possible situation — these cases are described in literature sources [44].
Currently, the correlation of seroma/complication notions is based on the criteria proposed by Morales-Conde [45]. The author proposed his work for laparoscopic surgery, but all its parameters are applicable to open surgery (Table 1).
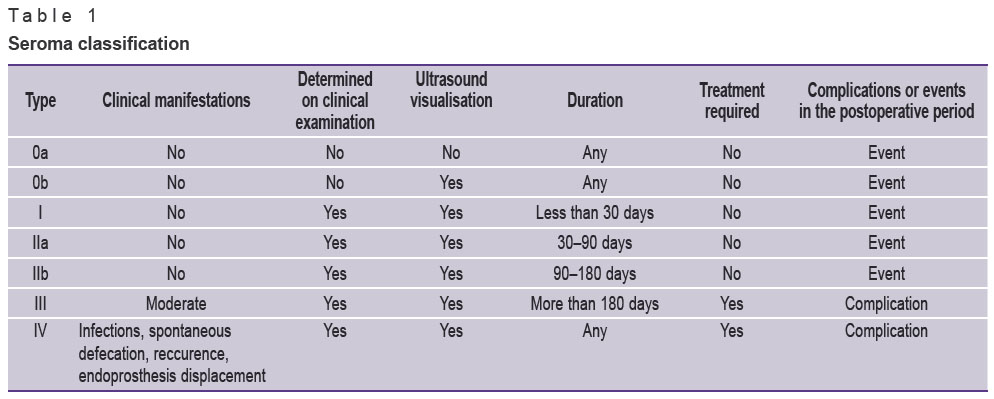
|
Table 1. Seroma classification |
In the last years, special classifications of events and complications in the surgical area have been created, they consider peculiarities of groups of patients and methods of surgery. For the patients of a herniological profile the classification was developed in detail by Petro and Novitsky [22]. The authors specify 4 categories: infections, sterile fluid collections, wound dehiscence, and an enterocutaneous fistula. The first category involves such concepts as cellulitis and infection of the surgical site (superficial, deep, organ or cavity). Seromas and hematomas are referred to the second category. The third and fourth categories were not specified in detail in the original work. It is worth mentioning that the above-mentioned classifications are completely different from the point of view of Petro and Novitsky and Morales-Conde and do not have principal contradictions.
The concept of infarction of subcutaneous tissue deserves special attention, it is a relatively new concept introduced by the leading Russian scientist in surgery of the abdominal wall hernias, Belokonev. According to his classification of wound events after herniotomy (2005), there are early and late complications. The first ones are the following: necrosis of wound edges, seromas (long exudation) infarction of the subcutaneous tissue, hematomas and purulence [46, 47]. To the second group the following complications are referred: implant cysts, paraprosthetic fistulas, mesh rejection, mesh contraction, implant edge detachment. Hernia recurrence, which is referred to the latter group, should still be regarded as an outcome, but not a complication. It was also convincingly proven that blood circulation impairment in one of the layers, which is located above the surface fascia and is supplied with blood from three rows of perforating blood vessels, causes infarction of the subcutaneous tissue [47].
The stage of an infectious complication and the state of the implanted mesh are of significance when the strategy of treatment is selected. The classification that takes into account all the mentioned factors was developed by Obolensky et al. [48]. The authors divided three types depending on the time passed from the reconstruction surgery (I — up to 30 days; II — from 1 month to 1 year; III — over 1 year) and three categories (S — stable, not deformed mesh; D — deformed but stable implant; N — endoprosthesis migration) [48, 49].
To define the risk of purulent and septic complications the class of a wound is taken into account. Here most frequently the classification of the American Center for Disease Control (CDC, Atlanta) is used [50]. A detailed stratification of patients with hernia in regard to the risk of complication development has also been proposed and is used now. Ventral Hernia Working Group — VHWG (2010–2012) also recommends dividing wounds into three types (low-risk, comorbid, potentially contaminated and infected) [22]. An improved classification (VHWG — Novitsky, 2016) includes only three types of wounds (risk, comorbid, contaminated) with specification of the last one: a (clean-contaminated), b (contaminated), and c (active infection) [22].
These authors suggested the newest approach (HPW, where H stands for hernia, P — for patient, W — for wound) that allows considering specific clinical cases in terms of risks of complications as well as measuring the risk in cohort studies. The first criterion: H1 — hernial orifice width is up to 10 cm; H2 — up to 20 cm; H3 — more than 20 cm. The second criterion: P0 — patient without comorbidity; P1 — morbid obesity, diabetes, smoking and/or immunosuppression. The third criterion takes into consideration the state of the surgical site: W0 — clean wound; W1 — contaminated. These authors systemize the data in the following way (Table 2).

|
Table 2. Stratification of hernia patients according to the risk of purulent and septic complications |
This approach is well-correlated with the finding of other researchers. It is known that patients with H2–3 hernias require specific and rather technically complex methods of surgery. According to Parent et al. [50] from 12 to 19% of these patients need repeated operations.
The modern authors emphasize an important role of using accurate terminology and concepts, specifying their meaning and use [51]. They suggest differentiating the following concepts: surgical site infection (SSI); surgical site occurrence (SSO); surgical site event (SSE); surgical site occurrence requiring procedural interventions (SSOIP) [51]. This approach has found application in recent studies [52].
Diagnostics of complications of prosthetic repair
Modern researchers first and foremost suggest identifying categories of risk where the development of complications is most probable. The proved criteria are obesity, smoking, previous surgical interventions and paraprosthetic infection in the medical history [53]. Early spotting of the postoperative problems of there are any is likely to be effective and in demand, but a successful diagnostic search will mostly determine the anticipation of the therapy outcome. From this point of view we should identify a special group of patients who already have ventral and incisional hernias with reduction of the abdominal cavity volume as well as a group of patients with an active infectious process in the future surgical site (H2–3, W1) with estimated maximal morbidity [22]. Here we can also observe the longest hospitalization, the highest morbidity, and mortality [54].
Technical difficulty of the operation and considerable load of the surgical site with synthetic material are associated with rather high reliability (minimal recurrent hernias frequency), but high rate of complications as well. For instance, according to the work [23], the patients operated with a sandwich technique with a double mesh implantation had no recurrent hernias, but in 33% of cases they had seromas and in 39% the patients needed surgical intervention due to an infection in the surgical site.
The reference literature provides various data on the effect of the previous purulent process caused by the undergone prosthetic repair on frequency of recurrent hernias. Some authors did not see any reliable impact of inflammatory problems in the medical history on the surgery outcome [55], other, however, demonstrated their absolute and determining significance — up to 67% of purulent complications in some groups of patients [23]. Apparently, it is the current activity of the infections in the surgery site that matters.
Anticipating wound complications requires a rather complex and multi-factor analysis [56, 57]. The duration of wound exudation has a strong correlation with the duration of hernia-carriage, the width of hernia orifice, the mesh square area, the endoprosthesis density; a number of other parameters are also important [57]. Some authors stress the influence of hernia sac volume, the presence of obesity in the patients and the importance of surgical access length [58]. Specificity of subcutaneous tissue structure in the para-umbilical area is under study, therefore tissue dissection direction can be of importance here [58]. Cardiovascular pathology and concomitant localization of hernias (medial segment and lateral location) are already confirmed risk factors of fluid accumulation [58]. The following factors also pose risk: female gender, large hernia orifice, surgery that lasts more than 2 h, diabetes mellitus, obesity, CHD and ischemic heart failure, low protein level [59].
Among other diagnostics methods along with general clinical ones the following methods of medical visualization play a certain role: ultrasonography [43, 60–63], computer and magnetic-resonance tomography [38, 63]. Thermography is also of importance as it can confirm the fact of an inflammatory process present and specify its location [64]. However, one should treat the findings on the abdominal pressure dynamics provided by this work skeptically. Despite the reliable difference shown, all the values of this parameter in the groups analyzed by the authors do not leave the reference interval (norm). The same researchers also highly appreciate possibilities of thermography in their further works [65].
Biochemical methods including the ones investigating the level of acute-phase proteins can be used as indicators of inflammatory complications. Screening of C-reactive protein as a parameter of injury rate of surgical intervention is well-known as it allows an objective assessment of various surgeries [66]. Russian authors used the methods of identification of ferritin and lactoferrin concentrations in wound exudate successfully [67]. This work shows that in the development of purulent complications their values were considerably higher. To assess a systemic inflammatory response after prosthetic repair it is recommended to determine the level of interleukins 1β, 6, 8, 10, vessels growth factor and tumor necrosis factor [68, 69]. According to the dynamic of concentrations of the given substances, scientists single out groups of patients with difference adaptation capacity. It is crucial for forecasting of the development of wound complications after prosthetic repair [70]. Double increase in the level of CCL2 chemokine and no decline in this value can verify the process of seroma formation [43]. In some cases serious complications (mesh migration into the abdominal cavity) do not have specific clinical manifestations and turn out to be unexpected [31].
Modern methods of treatment of purulent and septic complications of prosthetic repair
There is no unified and generally accepted approach to the treatment of infectious complications associated with a mesh implantation so far [71]. It is necessary to consider all the existing methods taking into account their benefits and drawbacks.
Puncture drainage under ultrasound control is the main and generally accepted method of treatment of the complications considered [43, 47, 60, 61, 72]. A freehand technique is most commonly used. In a number of clinics surgeons developed and successfully use original devices for puncture drainage as in their opinion it reduces surgical procedure injury and increase the effectiveness [73]. Suggested by the colleagues idea of infusion of chlorhexidine bigluconate solution into the cavity is of special interest. However, it is difficult to agree with the formulation of the device function, which is “for opening cavities”, because the authors obviously perform a puncture. We would also like to argue about the indications for use provided by the authors of this article [73] (accumulations with the volume of more than 4 cm3 and in the same publication — 40 mm3), in their further works [74] they are 4 cm3, because nobody else in the literature available describes such small-volume accumulations. It is absolutely obvious that the latter ones resorb spontaneously and do not have any clinical and practical significance. Modern authors emphasize that small volume of exudate in the mesh implantation site can be observed in practically all patients [43, 59]. Most of these accumulations resorb on their own, only in some situations the presence of significant volume of fluid can result in complications [59].
Surgical strategies in case when there are already developed purulent complications associated with a mesh can be different [75]. Some of them are rather difficult to identify during some research description, because there is no universal classification of the methods of following-up patients with paraprosthetic infection. The first variant includes complete removal of the infected implant (in a number of cases — dissection with the surrounding mesh tissues) with simultaneous reconstruction/correction of the abdominal wall and implantation of another endoprosthesis. The second variant presupposes only removal of the mesh (complete or partial) and delayed reconstruction. The third one involves the strategy of preservation (saving) of endoprosthesis left by the surgeon in situ, treatment of pyogenic infection with systemic and/local antibacterial therapy and special technologies targeted at sanitation of the suppurative focus. It is possible to combine these strategies or use than consequently.
Surgical intervention with removal of the implanted endoprosthesis. Experience in removing previously implanted meshes in most clinics is limited due to some objective reasons: such serious complications are rather rare in clinical practice. However, a number of researchers collected significant material that allows a comprehensive analysis and proper conclusions. Sharma et al. [76] for 4.5 years of practice removed 105 previously implanted meshes. Infection was one of the most frequent causes of this surgical intervention. Surgical interventions with mesh removal and simultaneous repeated prosthetic repair are referred to categories 3 or 4 according to the classification of Petro and Novitsky [22]. In a number of cases endoprostheses of biological origin are recommended for these purposes. But the frequency of unsatisfactory results after such interventions is unacceptably high, namely 57.5% of wound complications, 85.7% recurrent hernias after bridging repair and up to 40% of those after other variants of closure of the abdominal wall defects [75]. In most cases (68%) endoprostheses placed onlay had to be removed [7, 8].
Some authors are proponents of early dissection of the implant in case it is infected [77]. However, the term “early removal” has not been clearly defined; the publication considered describes experience of treating patients with formed external fistulas that existed from 1 to 6 years. The researchers performed complete dissection in 8 out of 10 patients, in the other 2 the mesh was explanted partially; the abdominal wall was repeatedly replaced with prosthesis in 4 patients, and 1 patient had the same complication again [77]. Authors are unanimous that the presence of chronic paraprosthetic infection is an indication for the implant excision.
Also an indication for mesh removal is damage of the hollow organ wall with an endoprosthesis [33]. Other foreign researchers are of similar opinion: in case of chronic, late symptomatic paraprosthetic infection meshes are most commonly to be removed. The decision is made after two weeks of fruitless attempts of conservative therapy [78]. It should also be taken into account that complete removal of endoprosthesis in a number of cases is technically complicated and sometimes impossible due to evident invasion of a reticular (woven) mesh in the connective tissue even under infectious conditions [79]. To make the procedure easier Russian authors recommended using ultrasound cavitation [80–82]. For mesh excision the authors used a titanium waveguide in the medium of water solution of chlorhexidine bigluconate combing physical and pharmacological effects. The possibility of delicate, low-trauma extraction of the implant from the surrounding tissues is a matter of special attention [80].
A choice of methods of complete or partial removal of a mesh has not been clearly defined yet. Levy et al. [71] demonstrated that complete mesh removal frequently led to complete infection management, but some patients who underwent partial explantation needed repeated surgery aimed at extra complete or partial excision of endoprosthesis fragments. However, the same study noted that after partial dissection of the implant, as a rule, there are no recurrent hernias, but after complete dissections the frequency of recurrent hernias is considerable [71]. Taking into account the risk of possibly needed prosthetic surgery in the previously contaminated surgery site it is rather problematic to define the tactics (whether to remove the endoprosthesis completely or not). Chung et al. [13] presented a detailed treatment analysis of 15 patients with chronic infection of endoprostheses and median manifestation of a purulent process for 15 months (7–49). The patients who had undergone complete mesh removal later had less purulent fistulas, but the author did not notice considerable differences in the number of recurrent hernias.
Explantation of the infected mesh and repeated prosthetic repair of the abdominal wall can be done simultaneously or step by step. It should be taken into account that implantation of synthetic material in the infectious conditions can be accompanied by high frequency of recurrent hernias (50% and more). According to some researchers, the second variant can reduce the frequency of infections in the surgical site up to 27% [83]. On the other hand, successful experience of explantation of infected meshes and simultaneous prosthetic repair of the abdominal wall in 41 patients was described in detail by Birolini et al. [7]. The authors came to the conclusion that removal of the infected endoprosthesis with simultaneous prosthetic repair of the abdominal wall and implantation of a polypropylene mesh led to successful outcomes in 95% of patients with chronic paraprosthetic infection. It turned out to be possible in conditions of severe contamination and active infection and the frequency of recurrent hernias was 34%, frequency of surgery due to surgical site infection (SSI) was 12.2%, and hernia recurrences constituted only for 4.9%. In the further study [8], which included 22 patients undergoing explantation/repeated prosthetic repair under conditions of MRSA infection, the authors confirmed the validity of their approach. The given results should be referred to the best ones in the category of patients under consideration, although it is difficult to agree with the onlay method of repeated prosthetic repair chosen by the authors [7].
According to the analysis of the published findings we can suppose that a chronic purulent process associated with an endoprosthesis is an indication for surgery. The scale of intervention is possibly complete dissection of the infected mesh. The issue of repeated prosthetic repair of the abdominal wall, the possible tactics and time is to be studied further. In meta-analysis [84] the following features that can predict the risk of mesh infection were reliably determined: administration of immunosuppresors by the patient, urgent interventions and SSI after reconstruction. The need for further mesh removal is clearly associated with the use of polytetrafluoroethylene endoprosthesis, implant location in onlay position and opening the intestinal lumen during surgery (resection and unintended enterotomy) and was reliably confirmed by other studies [85, 86].
Strategy of saving the mesh and negative pressure wound therapy. Negative pressure wound therapy (NPWT) was originally suggested and implemented Argenta and Morykwas. It was used for treating patients with purulent wounds, peritonitis, but soon it started to be successfully used in herniological clinical practice [87]. The national researchers used it to reduce the number of cases of mesh removal in patients with purulent and septic complications associated with prosthetic repair from 33.5 to 13.5%, and the frequency of infectious process recurrences from 22.2 to 17.3% [49].
Foreign authors analyzed the experience of 722 prosthetic repair with 233 (30.3%) of wound complications, including 48 patients who underwent NPWT, all the meshes were left in situ [87]. In average 9 replacement sets (wound dressings) were required, the average therapy duration was 34 days. The scientists [10] came to the conclusion that paraprosthetic infection can be successfully stopped with negative pressure systems, and NPWT must be considered an obligatory procedure of the first-line therapy of this complication. In their work [10] the duration of the method was 199 days in average (including 27 days in hospital); the dressings were changes three times a week. Other researchers [88] describe experience of treating 63 patients with NPWT (52 after retromuscular reconstruction and 9 after intraperitoneal reconstruction) that lasted 34 and 62 days respectively in conditions of Staphylococcus aureus, Escherichia coli or Klebsiella strain infections. In all the cases macroporous polypropylene meshes were saved. The authors used pressure of –80 mm Hg, and in cases of intraperitoneal endoprosthesis placement it was –60 mm Hg, then (after granulation appearance) they also used –80 mm Hg.
A possibility of intraperitoneal mesh placement in combination with NPWT was also studied in regard to wound closure in patients who underwent laparotomy [89]. In this case lower negative pressure values were used (–25 to –75 mm Hg). They published the data on successful in situ preservation of a titanium mesh implanted onlay under conditions of MRSA associated active process [90]. The experiment [91] stated that presence of a mesh in the surgical site does not make any difference to the course of the inflammatory process if it was placed lege artis. However, its deformity (twisting) actively promotes the colonization with microorganisms.
Taking into account the presented data we can draw a conclusion that essential conditions of using the strategy of mesh preservation are as follows: acute inflammatory process, macroporous and monofilament structure of the endoprosthesis, NPWT availability and experience in this therapy, a possibility of adequate microbiological monitoring and targeted medication control of the infectious process, lack of technical errors in the implantation performed with preferable placement of the endoprosthesis in the retromuscular space. In this situation the patient can be treated without the implant removal.
A possibility of effective NPWT use in case of deep seromas and excessive lymphorrhea via the wound was noted by foreign authors [92]. Some surgeons used the pressure range from –100 to –150 mm Hg. In the same way NPWT can be used for treatment of extensive infected seromas. For sooner obliteration of the cavity the authors used doxycycline as a sclerosant [93].
The use of physical factors and based on their possibilities methods are actively developing. It was shown that the pulsing electrical field can destroy a biofilm and eradicate one of the most problematic microorganisms — Pseudomonas aeruginosa [94].
Prevention of purulent and septic complications of prosthetic repair
The reference literature describes in detail a set of measures targeted at SSI prevention, which is a combination of organizational and applied solutions including the rules of aseptic, antiseptics and pharmacological suppression of the current flora with highest evidence level [95–97]. A part of these measures are observed everywhere, but some of them — only in a number of clinics where it is possible. It was proved that in inpatient clinics that carry out a significant number of herniological operations the frequency of serious complications is reliably lower, although the duration of treatment and its cost are higher [98].
One should also consider the rules which were proposed by American surgeons and have the highest evidence levels and can be applied in elective surgery [99]. In their opinion it is necessary to carry out mesh reinforcement, but not bridging repair, and use laparoscopic methods if there are indications. To reduce the frequency of infections associated with prosthetic repair they recommend not operation on patients with body mass index more than 35 kg/m2, with the level of glycosylated hemoglobin of 8% and higher as well as current smokers. It does not mean complete rejection from surgery, but a surgeon should avoid operating on this category of patients if possible. Patients are recommended to refuse from smoking and to address to specialists for normalization of body weight and glycemia.
Diabetes mellitus in herniological clinical practice is reliably recognized an independent predictor of complications [100]. Optimization of body weight proved to reduce the costs of treating patients with hernia [101]. Technically during the operation techniques should be implemented to complete the intervention precisely by reconstruction of the abdominal wall. Presence of MRSA infection is not also a straight contraindication, but it requires targeted SSI prophylaxis and surgeons’ readiness for complications and their treatment [9]. Surgical resection on the intestine can be also regarded as a risk factor [85]. In high-risk patients with maximum contamination of the surgical site and in patients with sepsis a step-by-step approach to treatment seems to be the most reasonable [83, 102].
As for prosthetic repair caused by hernias, modern scientists divide all the prevention methods into three groups: preoperational, intraoperational, and postoperational [59, 62]. One should also differentiate tactical aspects, technical solutions, medication, and hardware-based methods.
Reviewing a combination of tactical and technical solutions targeted at the abdominal wall reconstruction it should be mentioned that the character of tissue reaction to prosthetic repair is determined not only by the material of the latter and the layer or cavity selected for implantation, but also by a tension factor [103]. It was convincingly proved that it is the tension factor that defines the correlation between the main populations of cells in the area of mesh fibers which radically changes the known parameters of biocompatibility of the endoprosthesis with the specific properties and parameters. Modern experimental studies demonstrated that tension results in lipomatous transformation of the muscles of the medial segment, and in lateral groups fibrosis is observed in the same situations [103]. Clinical studies with detailed stratification of herniological profile patients depending on risk degrees and operations depending on their injury rate reflect that preference of non-tension methods is associated with low frequency of complications [104]. In the most complicated situations (operations on recurrent hernias) this approach is accompanied by the best results [105].
At pre-operational step, the key role in prevention of infections in the surgical site belongs to antibacterial prophylaxis which is carried out in accordance with generally accepted approaches [95, 96]. High load of the surgical site with synthetic material makes prevention requirements required. As it was recommended in the work [106], a single dose of the medication is sufficient. A number of Russian authors are of the same opinion [107].
Optimal medications for this purpose are cephalosporins of the 1st and 2nd generation; it is also possible to use protected penicillins or fluoroquinolones. Medications of other classes are used for targeted prevention, if the patient’s actual microflora is known. The optimal time of their administration depends on pharmacodynamics of the chosen medication: 30 min before the incision (betalactams), 60 min before (fluoroquinolones) or 2 h before (vancomycin). The use of cephalosporins of the 3rd generation is not recommended. These opinions are based on high-level evidence provided in meta-analyses [108]. It is shown that prolongation of antibacterial therapy does not affect the frequency of wound complications [109]. Foreign manuals emphasize that there is no significant difference in effectiveness among various medications [106], in some national studies [107] the selection of antibiotics is determined by pharmacoeconomic reasons.
The choice of operation methods and surgical techniques. There is a widespread idea that after laparoscopic variants of prosthetic repair of the abdominal wall the frequency of wound infections is lower than in case of open surgery [110]. This regularity is known not only in herniology, but it should be taken into account that the results of comparative analysis impose a number of limitations, firstly, the fact that not all patients can be effectively and successfully operated on with laparoscopic approach. Other aspects are connected with various variants of endoprosthesis placement in the abdominal wall layers (in most laparoscopic operations the mesh is implanted intraperitoneally [33].
However, currently, the technology of open retromuscular prosthetic repair is so much enhanced, standardized and practically developed that the above-mentioned differences in the results are not absolutely true. The meta-analysis [21] demonstrated that there are fewer complications after open retromuscular reconstruction than after the laparoscopic intraperitoneal one. The mesh has significant drawbacks from the point of view of intraoperational complications, such as bleedings and intestinal damage. No significant differences in hernia recurrence and pain frequency in one-year follow-up period were registered. The studies of Belyansky et al. [111] are of great interest regarding the combination of advantages of retromuscular technique and endoscopic access. They proposed eTEP/eTAR operations performed only with an endosurgical method with an extraperitoneal access with 3.8% of wound events and no infectious complications per 79 and there were similar results in the series of 37 robotic eTEP/eTAR [112].
During an open surgery some targeted surgical techniques can contribute to risk reduction in purulent-septic complications. A tactic solution to refuse from onlay reconstruction will be the most justified of all. It has been proved that separation of cutaneous-subcutaneous flaps along with subcutaneous tissue contact with an endoprosthesis is the breeding ground for infections in the surgical site. That is why the onlay method is not recommended for routine herniological practice. It can be used only as an exception.
According to the data of meta-analysis [1], for onlay technique up to 31% of wound complications are typical. In other studies this parameter was 21–42% [113]. It is no surprise that in some modern manuals for treating patients with incisional hernias this method is not even described [114]. If authors use this variant of prosthetic repair, they use special techniques that allow fixing the cutaneous-subcutaneous flap to the adjacent structures, mesh, aponeuroses [60]. In that work it could reduce the frequency of wound complications from 41.9 to 17.3%. Similar solutions aimed at elimination of residual cavities in the wound were suggested by the colleagues [115]. Other researchers stress that the most important factor of complication prevention is minimal mobilization of cutaneous-subcutaneous flap from aponeuroses, therefore it is better to refuse from onlay technique [116].
Belokonev’s method [105] also involves separation of flaps and presupposes the contact of a mesh with subcutaneous tissue, but it differs significantly from the standard onlay technique and is used successfully in the most difficult cases. In the original method the mesh is placed not on the aponeuroses, but on abdominal rectus muscles which are well blood-supplied, can actively resorb wound exudate and are the source of angiogenesis in the area of reconstruction and the growth of connective tissue around the mesh fibers. The standard onlay technique does not have these advantages.
Some authors refuse from complete isolation and dissection of a hernia sac preferring desquamation of its mesothelium and intraperitoneal mesh placement [117].
When treating patients with large hernias one should pay special attention to the choice of the separation technique of the components in situations with high risk of development of infectious complications. The greatest personal experience belongs to American surgeons: 700 anterior components separation technique (ACST), including 311 ones which were analyzed in detail [118] and 428 consequently performed transversus abdominis release (TAR — transversus abdominis release) [119]. They and the authors of the following works [120, 121] demonstrated the advantages of an endoscopic ACST variant (11–20.6% of wound complications) before Ramirez technique (33–34.6%), as well as TAR method (18.7%). However, in further meta-analyses [122, 123] those differences were not confirmed. Frequency of events in the surgical site was 21.4% after anterior components separation technique, 23.7% after TAR, 20.3% during ACST with an endoscopic technique and 16% after anterior separation with preservation of perforating vessels [124]. Preference of this or that variant of component separation remains at the discretion of the operating team depending on their experience, possibilities and the patient.
A number of technical solutions used in postoperative period can prevent SSI development. Prevention with NPWT is not well described in literature. In the works of the Russian authors [125] 24 h after the operation for 7 days daily an original device from vacuum therapy was used with the following parameters: pressure –120 mm Hg, exposition — 15 min. It could reduce the number of indications for traditional drainage and reduce the frequency of complications. This method proved its reliability in the experiment and the findings were confirmed morphologically [126]. The researchers proved that duration of the exudative phase of an inflammatory process at the NPWT background reduced considerably.
In this work [127] the scientists used pressure –100 mm Hg continuously on the closed wound for 5 days, then they evaluated the state of the operation site. The procedure was canceled when there was no edema or risk of wound dehiscence, in it was needed the procedure was prolonged for 3–5 days more. Patients who underwent a complex reconstruction of the abdominal wall took part in the study. Operations lasted more than 400 min, practically in all the patients were exposed to CST (components separation technique); it can help evaluate the scale and complexity of surgical intervention. The work showed reliable, practically double reduction in the number of purulent complications, especially the superficial access infections (in 6.8 times). The method did not affect the risk rate of intra-abdominal complications, the need for repeated hospitalization, frequency of repeated laparotomies, development of hematomas, seromas, and necrosis of cutaneous wound edges. This method has the following undoubted advantages: non-invasiveness, technical simplicity, availability in many clinics, skills, and experience in work with modern vacuum systems accumulated by surgeons during the treatment of patients with purulent wounds and peritonitis. The analytical review of Massey [128] showed the reduction in frequency of seroma formation from 11.3 to 6.4% when NPWT was used, but these differences were not valid according to the meta-analysis data.
Drainage installation after prosthetic repair is a routine measure approved by the majority of surgeons. However, correlation between the anticipated benefit (reduction in the frequency of hematomas, seromas) and the potential risk (entrance gate for infections) is not as favorable as it may seem at first sight. In the meta-analysis [129] the authors proved that there are not enough convincing data on a positive effect of drainages. In the up-to-date randomized cohort controlled study [130] there were assessed the results of drainage after retromuscular reconstruction. It has been demonstrated that the use of drainages does not affect the frequency of events in the operational site, formation of hematomas, all types of SSI, necrosis of cutaneous-subcutaneous flaps, separation of wound edges and dehiscence of fascia structures. In this study seromas were observed much more rarely in case drainages were in situ.
Russian researchers [61] use drainages during the operation and perform pre-peritoneal irrigation with ozonized 0.9% solution of sodium chloride and 2.5% solution of Ropivacaine of 20 ml each 6–8 h for 2–3 days. The authors explain positive results with stimulation of reparative processes and microcirculation improvement. Representatives of other surgical schools successfully use infusion of water solution of chlorhexidine (15 ml) into serous cavities right after evacuation of the exudate; it helps eliminate the development of pyogenic infection [73, 74].
The work [16] stated in the experiment that the use of diclofenac and ibuprofen effectively prevents the formation of Staphylococcus aureus and Escherichia coli biofilm on the surface of polypropylene mesh; obviously, these medications inhibit adhesion of bacteria to the endoprosthesis fibers.
The choice of an endoprosthesis. This step is important in terms of risks of possible pyogenic infection associated with the implant. There are more classifications of meshes; and the most interesting and topical ones were analyzed by Anurov in his review [131]. Depending on the production technology, construction properties, peculiarities of integration in the abdominal wall tissues and the area of application the author specifies the following five types: reticular, membrane, composite and 3D (volumetric), emphasizing special possibilities of the 4th type in case of infections. Most authors do not object the preference of macroporous meshes to microporous ones which are less resistant to infections. The latter are no longer used in high-risk patients and in conditions of the surgical site contamination. It was proved that the key role belongs to the correlation of pore size, microorganisms (1–2 µm), macrophages (18–35 µm) and leucocytes (15–20 µm) [15]. The researchers state that when pore sizes are less than 15 µm, infectious agents easily penetrate into them and cannot be attacked by immunocompetent cells.
From numerous materials the material that is the most resistant to infections is polypropylene fiber due to its solidity and hydrophobicity [15]. Polyvinylidene fluoride has similar, but in some relation better properties for a mesh material. The experiment [132] stated that composite endoprosthesis with an adhesion barrier from collagen should not be used in conditions of bacterial contamination. Such cultures as Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Staphylococcus aureus demonstrate continuous lawn growth on the adhesive membrane and under it. Polytetrafluoroethylene (ePTFE) is a classic example of the material, application of which is associated with considerable frequency of infectious complications and therefore it not recommended for use under contamination conditions [13].
The use of materials of biological origin is the subject of scientific debate, although in the recommendations of the World Society of Emergency Surgery (2017) on surgical treatment of patients with strangulated hernias it is clearly said that in the 1st and 2nd class of wound according to CDC classification it is recommended to use prosthetic repair (1A level of evidence) [133]. And the frequency of wound complications is not increased (wound class 1), 30-day frequency of complications associated with access does not increase (wound class 2). For wound classes 3 and 4 surgeons should refuse from prosthetic repair when the defect is less than 3 cm, and in other cases biological prostheses should be used (2C level of evidence), but if they are unavailable a resorbing mesh of polyglactin is applicable (2C level of evidence) [133].
The literature has evidence about the use of autologous skin flaps for closing the defect of the abdominal wall with onlay method (including the one with anterior separation) with SSI frequency of 13.6–20% [134, 135]; the biggest positive experience of such abdominal wall reconstruction was accumulated by Botezaty.
In most cases modern implants of biological origin have xenogeneic origin and represent acellular scaffolds from collagen fibers which are in some cases exposed to crosslinking [131]. They are gradually resorbed and replaced by their own connective tissue at these steps preserving certain strength, extensibility, and resistance to infection. However, positive properties of endoprosthesis of biological origin in terms of resistance to infections have been disputed in a range of publications for the last 5 years. López-Cano et al. in the controlled cohort study [136] compared the results of abdominal wall reconstructions with biological and synthetic meshes under contamination conditions, did not determine advantages of biological ones in regard to complications and identified reliable (practically fourfold) differences in the recurrence rate in favor of synthetic meshes. The authors of two-centered clinical study [137] informed about 74% of complications in patients exposed to biological meshes under contamination conditions (VHWG classes 3 and 4). Those were the patients of classes 1–3 according to the classification of the American Society of Anesthesiologists (ASA) with relatively small square area of defects (143 and 146 cm2 in two groups respectively), but the majority of patients had intestinal stomas or infected endoprosthesis in the abdominal wall. Complications of class III–IV according to Clavien–Dindo were registered in 60% of patients that underwent bridging repair operations and in 28% of patients after reinforced repairs. It is worth mentioning that in 84% of cases the mesh was placed intraperitoneally [137].
Other authors inform about the use of biological endoprosthesis for reconstruction after a synthetic mesh removal, and the frequency of wound complications reached 48%. Furthermore, frequency of recurrence occurred later was also unacceptably high [138]. According to the meta-analysis [139], the results of using biological meshes in conditions of infections are not better than the results of using synthetic endoprostheses. Ferzoco [140] demonstrated that frequency of infectious complications after implantation of biological endoprostheses in wound classes 2–4 is 20–51%, and the need for further mesh removal ranges from 0–23%. The author emphasizes that there are not enough high-level studies that directly compare the results of implantation of biological and synthetic endoprosthesis in conditions of surgical site contamination. However, these works have recently appeared.
The study [141] demonstrated that biological endoprosthesis in the situations of potential contamination do not have advantages over the synthetic ones (21 and 12% SSI respectively), and in contaminated conditions the results are even worse (38 and 11%). Sahoo et al. [52] proved on wide material (8005 patients operated on ventral and incisional hernias) obtained from the detailed analysis of the database of the American Society of Herniologists that SSI frequency after implantation of biological endoprosthesis under contamination was two times higher than after the use of synthetic meshes. In the latest publication with 1A level of evidence Köckerling et al. [142] presented a detailed analysis of a number of works on this topic and proved convincingly that currently accumulated data referring to the use of biological and biosynthetic meshes for operating on patients with the abdominal wall hernias under conditions of infection do not confirm the advantages of biological endoprosthesis in the same conditions. American authors proved on wide material (1023 patients) that the use of biological endoprosthesis is a predictor of complications [99] and higher costs of the patients’ treatment [100].
There is an impression that the issue subjected to such a detailed and effective analysis in the recommendations of the World Society of Emergency Surgery dated 2017 is to be critically considered and reviewed. There are few studies on the use of resorbing meshes in contamination conditions; their authors revealed the same regularities as for non-absorbable endoprosthesis. As for the body reaction, resorption process, thickness of the reparation zone, strength and bacterial colonization, more stable and favorable results were obtained for monofilament macroporous endoprosthesis compared to multifilament microporous endoprosthesis [143]. However, hernia recurrence rate after the use of absorbable implants is inappropriately high; and it reasonably and considerably limits their administration to temporary closure of the abdominal cavity, for example, after laparostomy.
Implantation prevention of infections in the surgical site. Some producers of endoprostheses made certain attempts to create meshes which have maximum resistance to infectious process and achieved certain success [144, 145]. They described endoprostheses containing polymer coating with silver nanoclusters [62]. The studies in vitro proved that this construction prevents colonization of mesh surface and biofilm formation, similar findings were obtained in some works in vivo. A mesh of polytetrafluoroethylene indicated for intraperitoneal implantation with double-sided coating containing silver carbonate and chlorhexidine diacetate was highly assessed in the experimental study using the culture of Staphylococcus aureus 106 CFU/ml [145]. A polyester mesh processed with polymerized cyclodextrin with vancomycin loading was used in the experiment in vivo under contamination with MRSA, showed excellent results compared to the control group [146]. Similar studies are successfully done by other authors [147].
The following substances injected in the endoprosthesis, applied on its surface and included in the composition of its coating are known as components: polyethylene oxide, polyethylene glycol, polysilazane, cyclodextrin, gentamicin, vancomycin, ofloxacin, rifampicin, amoxicillin, ciprofloxacin, vancomycin, lysostaphin (endopeptidase), chlorhexidine, triclosan, quaternary ammonium salts, nitric oxide, silver, titanium, gold, and palladium [146–148]. It is worth mentioning that encouraging results were received only in the studies with synthetic materials. Modification of biological endoprostheses for the use under contamination with Escherichia coli in the experiment was performed in an absolutely different way (mesenchymal stem cells were used) and has not met the expectations yet [149].
To fix meshes to the abdominal wall tissues suture materials impregnated with antibacterial means were offered. These threads in various representations contain chitosan, germanium, organic compounds, fluoroquinolone or other components (doxycycline) [150, 151]. According to the experimental data, the use of these materials causes taxis of phagocytizing cell population in the surgical site and reduces duration of phase 1 of the wound process and reduces severity of local inflammatory signs [150]. In clinical conditions the use of biologically active suture materials (the authors call it implantation antimicrobial prophylaxis) contributes to the reduction of SSI rate [151].
Researchers do not position this approach as an alternative to standard antibacterial prophylaxis, but consider it as a supplementary intra-operational measure to prevent SSI and to be performed on surgical site of class 3 and 4. Wide material (840 patients with incisional hernias) showed that even the use of capron meshes with impregnation with macrolides can considerably reduce SSI rate [152]. The same authors proposed coating for a polypropylene endoprosthesis based on polyvinyl alcohol and cefotaxime; their experiment proved effectiveness of the method and investigated the course of reparative process after implantation of these endoprostheses [153].
The latest direction in the prevention of purulent and septic complications during prosthetic repair is preliminary processing of meshes with antiseptic and/or antibiotics ex tempore — right before the implantation. A number of reviews [62, 154], experimental works (in vitro and/or in vivo) [148, 155] and clinical studies referring to SSI prevention or including the data referred to it have recently been published [156]. Foreign authors used short-term exposure of the implanted meshes to 0.05% water solution of chlorhexidine bigluconate (variant 1) and to the same solution but including 900 µg/ml of allicin (variant 2) [148]. An antibacterial effect of this method was convincingly proved on agar in vitro. Variant 2 demonstrated the maximum diameter area of Staphylococcus aureus culture suppression. The controlled experiment on rabbits confirmed effectiveness of the method, and here the second variant of prevention was also the best [148]. It is remarkable that preliminary processing of an endoprosthesis did not interfere with the course of a reparative process.
Authors of another study [155] in vivo used preliminary soaking of implanted endoprostheses in vancomycin solution (10 mg/ml) for 10 min as part of targeted MRSA infection prevention. The work studied polyester multifilament meshes and monofilament polypropylene endoprostheses, composite and standard ones. All meshes made a decolonization effect. However, upon careful consideration of the results it is getting clear that the problem has not been completely solved. The use of antibiotics in all the cases did not prevent biofilm growth: 44–50% of surface of multifilament endoprostheses was covered with staphylococcus biofilm; the least colonized was a standard monofilament (non-composite) polypropylene mesh, but the difference of its values compared to other prostheses was not valid.
National scientists processed a mesh with 0.5% water-alcohol solution of chlorhexidine in clinical conditions [156], but the controlled findings on the effectiveness of this technique remained outside the scope of the publication. The authors previously studied an effect of chlorhexidine on the already formed biofilms of Pseudomonas aeruginosa and Staphylococcus aureus; a positive was described, but biofilms were not completely destroyed [157]. In the experiment processing with chlorhexidine and prontosan of the adhesion barrier of collagen as a component of a composite endoprosthesis with its further contamination with Escherichia coli, Klebsiella pneumoniae, Staphylococcus aureus cultures inhibited or slowed down microorganism growth, and in the experiment with Pseudomonas aeruginosa did not prevent colonization [132]. The obtained results can be applied in clinical practice only with some adjustments, as right after the placement of an endoprosthesis in situ local concentrations of antibacterial agents are decreasing fast as they are spread to the surrounding tissues [148].
Conclusion
On the basis of the analysis of the data from modern literature sources, we can draw a conclusion that the problem of infectious complications of prosthetic repair is currently far from being solved. Its further investigation at all steps requires a complex multidisciplinary approach based on clinical findings obtained in accordance with the concept of evidence-based medicine and the results of fundamental studies.
Planning of preventive measures should be started at the pre-operation step in accordance with an elective approach, thorough stratification of the patient, detailed evaluation of risks and an objective status of the patient. Antibiotic prophylaxis should be done in accordance with the international guidelines, but target solutions should comply with specificity of each individual patient: their up-to-date microbiota (local and outside the surgical site), medical history and the current state. Taking into consideration all the above-mentioned factors and measures it is necessary to choose a surgical method, and in accordance with it an appropriate endoprosthesis, giving preference to retromuscular mesh placement and the use of monofilament reticular meshes made of minimally colonized materials. In some situations during surgery it is possible to use simple and effective methods of topical preventions, including the preparation of endoprostheses ex tempore.
It is reasonable to treat paraprosthetic infections in centers that have big experience in this sphere, are equipped with a wide range of infection control tools and the surgical methods and techniques applied: from NPWT to explantation of an infected mesh with component separation, repeated prosthetic repair of the abdominal wall and mesh placement in the layers free from contamination and in the zones with a possibility of step-by-step and simultaneous surgery.
Research funding. The study was funded by the internal grant of the Privolzhsky Research Medical University.
Conflict of interest. The author declares no conflict of interest concerning the content of the article.
References
- Deerenberg E.B., Timmermans L., Hogerzeil D.P., Slieker J.C., Eilers P.H., Jeekel J., Lange J.F. A systematic review of the surgical treatment of large incisional hernia. Hernia 2015; 19(1): 89–101, https://doi.org/10.1007/s10029-014-1321-x.
- Vorst A.L., Kaoutzanis C., Carbonell A.M., Franz M.G. Evolution and advances in laparoscopic ventral and incisional hernia repair. World J Gastrointest Surg 2015; 7(11): 293–305, https://doi.org/10.4240/wjgs.v7.i11.293.
- Payne R., Aldwinckle J., Ward S. Meta-analysis of randomised trials comparing the use of prophylactic mesh to standard midline closure in the reduction of incisional herniae. Hernia 2017; 21(6): 843–853, https://doi.org/10.1007/s10029-017-1653-4.
- Jensen K.K. Recovery after abdominal wall reconstruction. Dan Med J 2017; 64(3): B5349.
- Hodgkinson J.D., Maeda Y., Leo C.A., Warusavitarne J., Vaizey C.J. Complex abdominal wall reconstruction in the setting of active infection and contamination: a systematic review of hernia and fistula recurrence rates. Colorectal Dis 2017; 19(4): 319–330, https://doi.org/10.1111/codi.13609.
- Petukhova I.N., Sokolovskiy A.V., Grigor’evskaya Z.V., Bagirova N.S., Tereshchenko I.V., Varlan G.V., Aginova V.V., Dmitrieva N.V. Infections associated with placement of foreign materials (prostheses, meshes, implants). Zlokachestvennye opukholi 2017; 7(3–S1): 57–60.
- Birolini C., de Miranda J.S., Utiyama E.M., Rasslan S. A retrospective review and observations over a 16-year clinical experience on the surgical treatment of chronic mesh infection. What about replacing a synthetic mesh on the infected surgical field? Hernia 2015; 19(2): 239–246, https://doi.org/10.1007/s10029-014-1225-9.
- Birolini C., de Miranda J.S., Utiyama E.M., Rasslan S., Birolini D. Active Staphylococcus aureus infection: Is it a contra-indication to the repair of complex hernias with synthetic mesh? A prospective observational study on the outcomes of synthetic mesh replacement, in patients with chronic mesh infection caused by Staphylococcus aureus. Int J Surg 2016; 28: 56–62, https://doi.org/10.1016/j.ijsu.2016.02.062.
- Hicks C.W., Blatnik J.A., Krpata D.M., Novitsky Y.W., Rosen M.J. History of methicillin-resistant Staphylococcus aureus (MRSA) surgical site infection may not be a contraindication to ventral hernia repair with synthetic mesh: a preliminary report. Hernia 2014; 18(1): 65–70, https://doi.org/10.1007/s10029-012-1035-x.
- Meagher H., Clarke Moloney M., Grace P.A. Conservative management of mesh-site infection in hernia repair surgery: a case series. Hernia 2015; 19(2): 231–237, https://doi.org/10.1007/s10029-013-1069-8.
- Narkhede R., Shah N.M., Dalal P.R., Mangukia C., Dholaria S. Postoperative mesh infection — still a concern in laparoscopic era. Indian J Surg 2015; 77(4): 322–326, https://doi.org/10.1007/s12262-015-1304-x.
- Filippou D. Late Ps. aeruginosa inguinal mesh infection 12 years after the initial operation: report of the case and short review of the literature. Case Rep Surg 2017; 2017: 4385913, https://doi.org/10.1155/2017/4385913.
- Chung L., Tse G.H., O’Dwyer P.J. Outcome of patients with chronic mesh infection following abdominal wall hernia repair. Hernia 2014; 18(5): 701–704, https://doi.org/10.1007/s10029-014-1277-x.
- Luhmann A., Moses A. Successful conservative treatment of a candida albicans intraperitoneal mesh infection following laparoscopic ventral hernia repair. Hernia 2015; 19(5): 845–847, https://doi.org/10.1007/s10029-013-1183-7.
- Zhukovskiy V.A. Polymer implants for reconstructive surgery. Innova 2016; 2(3): 51–59.
- Reśliński A., Dąbrowiecki S., Głowacka K. The impact of diclofenac and ibuprofen on biofilm formation on the surface of polypropylene mesh. Hernia 2015; 19(2): 179–185, https://doi.org/10.1007/s10029-013-1200-x.
- Zakhidova S.Kh. Mistakes and complications associated with tension-free hernioplasty of strangulated postoperative ventral hernias. Tyumenskiy meditsinskiy zhurnal 2014; 4(16): 55–56.
- Tashtemirova O., Abitanova A., Zhakupova G. Analysis of surgical treatment of ventral hernia for patients with obesity. Nauka i zdravookhranenie 2013; 6: 61–64.
- Tubre D.J., Schroeder A.D., Estes J., Eisenga J., Fitzgibbons R.J. Jr. Surgical site infection: the “Achilles Heel” of all types of abdominal wall hernia reconstruction. Hernia 2018; 22(6): 1003–1013, https://doi.org/10.1007/s10029-018-1826-9.
- Cherepanin A.I., Povetkin A.P., Lutsevich O.E. Atlas oslozhneniy khirurgii gryzh peredney bryushnoy stenki [Atlas of surgical complications of anterior abdominal wall hernias]. Moscow: GEOTAR-Media; 2017; 208 p.
- Köckerling F., Schug-Pass C., Scheuerlein H. What is the current knowledge about sublay/retro-rectus repair of incisional hernias? Front Surg 2018; 5: 47, https://doi.org/10.3389/fsurg.2018.00047.
- Petro C.C., Novitsky Y.W. Classification of hernias. In: Hernia surgery. Springer International Publishing; 2016; p. 15–21, https://doi.org/10.1007/978-3-319-27470-6_2.
- Köckerling F., Scheuerlein H., Schug-Pass C. Treatment of large incisional hernias in sandwich technique — a review of the literature. Front Surg 2018; 5: 37, https://doi.org/10.3389/fsurg.2018.00037.
- Ozaki K., Tanimura K., Ebina Y., Kanemitsu K., Yamada H. A pregnant woman with a surgical site infection after mesh repair of an abdominal wall incisional hernia: a case report. J Med Case Rep 2017; 11(1): 66, https://doi.org/10.1186/s13256-017-1217-3.
- Doran H., Costache A., Mustăţea P., Pătraşcu T. Enterocutaneous fistula occurring 15 years after the prosthetic mesh repair of a recurrent incisional hernia — a case report. Chirurgia (Bucur) 2015; 110(5): 478–481.
- Asano H., Yajima S., Hosoi Y., Takagi M., Fukano H., Ohara Y., Shinozuka N., Ichimura T. Mesh penetrating the cecum and bladder following inguinal hernia surgery: a case report. J Med Case Rep 2017; 11(1): 260, https://doi.org/10.1186/s13256-017-1435-8.
- Degheili J.A., Hosn M.A., Lakis M.E., Hallal A.H. Sigmoid to scrotal fistula secondary to mesh erosion: a rare complication of inguinal hernia repair in a patient on anticoagulation. BMC Surg 2015; 15: 94, https://doi.org/10.1186/s12893-015-0070-9.
- Liu S., Zhou X.-X., Li L., Yu M.-S., Zhang H., Zhong W.X., Ji F. Mesh migration into the sigmoid colon after inguinal hernia repair presenting as a colonic polyp: a case report and review of literature. World J Clin Cases 2018; 6(12): 564–569, https://doi.org/10.12998/wjcc.v6.i12.564.
- Dubbeling R.M., Ramesh K. Infected inguinal hernia mesh presenting as pseudotumor of the bladder. Indian J Urol 2013; 29(4): 345–347, https://doi.org/10.4103/0970-1591.120120.
- Ishikawa S., Kawano T., Karashima R., Arita T., Yagi Y., Hirota M. A case of mesh plug migration into the bladder 5 years after hernia repair. Surg Case Rep 2015; 1: 4, https://doi.org/10.1186/s40792-014-0004-2.
- Bazaev A.V., Goshadze K.A., Malov A.A., Yanyshev A.A. Migration of polypropylene mesh into the abdominal cavity after hernia repair of recurrent postoperative ventral hernia (clinical case). Vestnik novyh medicinskih tehnologij 2016; 23(1): 59–61.
- Aggarwal S., Praneeth K., Rathore Y., Waran V., Singh P. Laparoscopic management of mesh erosion into small bowel and urinary bladder following total extra-peritoneal repair of inguinal hernia. J Minim Access Surg 2016; 12(1): 79–82, https://doi.org/10.4103/0972-9941.169956.
- Koyama M., Miyagawa Y., Yamamoto Y., Kitazawa M., Suzuki A., Ishizone S., Miyagawa S. Surgery for chronic mesh infection occurred 10 years after sacrectomy — mesh resection and mesentric leaf repair: a case report. Int J Surg Case Rep 2017; 30: 215–217, https://doi.org/10.1016/j.ijscr.2016.10.057.
- Shrivastava A., Gupta A., Gupta A., Shrivastava J. Erosion of small intestine with necrotising fasciitis of over lying abdominal wall after expanded poly-tetrafluoroethylene mesh implantation: a rare complication after laparoscopic incisional hernia repair. J Minim Access Surg 2013; 9(3): 138–140, https://doi.org/10.4103/0972-9941.115381.
- Dinc T., Kayilioglu S.I., Sozen I., Yildiz B.D., Coskun F. Fournier’s gangrene as a postoperative complication of inguinal hernia repair. Case Rep Surg 2014; 2014: 408217, https://doi.org/10.1155/2014/408217.
- Merali N., Almeida R.A., Hussain A. A case report on management of synergistic gangrene following an incisional abdominal hernia repair in an immunocompromised obese patient. Int J Surg Case Rep 2015; 15: 99–102, https://doi.org/10.1016/j.ijscr.2015.07.034.
- Eriksson A., Rosenberg J., Bisgaard T. Surgical treatment for giant incisional hernia: a qualitative systematic review. Hernia 2014; 18(1): 31–38, https://doi.org/10.1007/s10029-013-1066-y.
- Babazhanov A.S., Akhmedov G.K., Obidov Sh.H. Means of optimization of operative therapy of postsurgical ventral hernia at patients with III–IV degree obesity. Nauka i mir 2015; 7(23): 137–139.
- Zykov D.V., Khalyastov I.N., Chegurov V.I., Ovcharov A.V., Karapetyan G.N. Large incisional hernia in a patient with diabetes mellitus and Crohn’s disease: a case report. Vestnik meditsinskogo instituta “REAVIZ”: reabilitatsiya, vrach i zdorov’e 2018; 1(31): 75–78.
- Kao A.M., Arnold M.R., Augenstein V.A., Heniford B.T. Prevention and treatment strategies for mesh infection in abdominal wall reconstruction. Plast Reconstr Surg 2018; 142(3 Suppl): 149S–155S, https://doi.org/10.1097/prs.0000000000004871.
- Arinchev R.S. Prognozirovanie i profilaktika oslozhneniy v otdalennom posleoperatsionnom periode u bol’nykh s priznakami displazii soedinitel’noy tkani. Dis. … kand. med. nauk [Prediction and prevention of complications in the remote postoperative period in patients with the signs of connective tissue dysplasia. PhD Dissertation]. Tver’; 2018.
- Degovtsov E.N., Kolyadko P.V. Seromas as a complication of surgical treatment of incisional hernias of anterior abdominal wall using mesh implants: current state of the problem. Novosti khirurgii 2018; 26(1): 96–102.
- Belokonev V.I., Ponomareva J.V., Pushkin S.Y., Melentieva O.N., Gulyaev M.G. Potential predictors and morphological aspects of seroma development after plastic surgery of the anterior abdominal wall hernia. Novosti khirurgii 2014; 22(6): 665–670.
- Salamone G., Licari L., Agrusa A., Romano G., Cocorullo G., Gulotta G. Deep seroma after incisional hernia repair. Case reports and review of the literature. Ann Ital Chir 2015; 86: S2239253X15022938.
- Morales-Conde S. A new classification for seroma after laparoscopic ventral hernia repair. Hernia 2012; 16(3): 261–267, https://doi.org/10.1007/s10029-012-0911-8.
- Babaev A.P., Guliaev M.G. Prevention of wound complications in patients with the implementation of prosthetic hernioplasty in a combined way. Aspirantskiy vestnik Povolzh’ya 2014; 5–6: 85–87.
- Sonis A.G., Grachev B.D., Stolyarov E.A., Ishutov I.V. Prevention and treatment of infection wound complications at prosthetic hernia repair. Rany i ranevye infektsii 2014; 1(2): 16–23.
- Obolenskiy V.N., Kharitonov S.V., Enokhov V.Yu., Ermolov A.A. Negative pressure wound therapy in comprehensive treatment of infected wound complication after tension-free hernioplasty. Khirurg 2015; 8: 25–32.
- Obolenskiy V.N., Enokhov V.Yu., Kharitonov S.V. Clinical efficacy of local negative pressure method for treatment of infectious wound complications after non-stretching hernioplasty. Meditsinskiy alfavit 2018; 9(346): 45–49.
- Parent B., Horn D., Jacobson L., Petersen R.P., Hinojosa M., Yates R., Wright A.S., Louie O. Wound morbidity in minimally invasive anterior component separation compared to transversus abdominis release. Plast Reconstr Surg 2017; 139(2): 472–479, https://doi.org/10.1097/prs.0000000000002957.
- DeBordJ., Novitsky Y., Fitzgibbons R., Miserez M., Montgomery A. SSI, SSO, SSE, SSOPI: the elusive language of complications in hernia surgery. Hernia 2018; 22(5): 737–738, https://doi.org/10.1007/s10029-018-1813-1.
- Sahoo S., Haskins I.N., Huang L.C., Krpata D.M., Derwin K.A., Poulose B.K., Rosen M.J. Early wound morbidity after open ventral hernia repair with biosynthetic or polypropylene mesh. J Am Coll Surg 2017; 225(4): 472–480.e1, https://doi.org/10.1016/j.jamcollsurg.2017.07.1067.
- Rhemtulla I.A., Fischer J.P. Retromuscular sublay technique for ventral hernia repair. Semin Plast Surg 2018; 32(3): 120–126, https://doi.org/10.1055/s-0038-1666800.
- Poruk K.E., Farrow N., Azar F., Burce K.K., Hicks C.W., Azoury S.C., Cornell P., Cooney C.M., Eckhauser F.E. Effect of hernia size on operative repair and post-operative outcomes after open ventral hernia repair. Hernia 2016; 20(6): 805–810, https://doi.org/10.1007/s10029-016-1542-2.
- Blatnik J.A., Krpata D.M., Novitsky Y.W., Rosen M.J. Does a history of wound infection predict postoperative surgical site infection after ventral hernia repair? Am J Surg 2012; 203(3): 370–374, https://doi.org/10.1016/j.amjsurg.2011.12.001.
- Vlasov A.V. The prediction of the probability of seroma development in case of endoprosthesis replacement of ventral hernias. Sovremennye problemy nauki i obrazovaniya 2013; 2. URL: http://www.science-education.ru/ru/article/view?id=8585.
- Golovin R.V., Nikitin N.A., Prokopev E.S. Prognosis of development of complications of injuries after com-bined allohernia plastic interventions for postoperative ventral hernias of median localization. Sovremennye problemy nauki i obrazovaniya 2014; 2. URL: http://www.science-education.ru/ru/article/view?id=12914.
- Vnukov P.V., Sheptunov Yu.M. The comparative strain characteristics of the subcutaneous tissue longitudinal and transverse paraumbilical wounds (experimental research). Vestnik eksperimental’noy i klinicheskoy khirurgii 2016; 9(2): 165–168.
- Cherkasov M.F., Khindikaynen A.Yu., Pomazkov A.A. Methods of diagnosis, prevention and treatment of complications of hernia repair. Astrakhanskiy meditsinskiy zhurnal 2016; 11(4): 50–64.
- Aleksandrenkov N.V., Mykhin A.S., Rebcovsky V.A., Leontev А.Е. Wound closure method at subaponeurotic plasty with polypropylene mesh of large postoperative ventral hernia. Novosti khirurgii 2013; 21(1): 88–93.
- Alishev O.T., Shaimardanov R.S., Makhmutova Y.R. Prevention of large and giant postoperative ventral hernia prosthetic hernioplasty postoperative complications. Kazanskij medicinskij zurnal 2013; 94(3): 417–420.
- Narezkin D.V., Sergeev E.V. Preventive methods of pyo-inflammatory wound complications in herniotomy of strangulated postoperative ventral hernias. Novosti khirurgii 2014; 22(6): 743–749.
- Salamone G., Licari L., Augello G., Campanella S., Falco N., Tutino R., Cocorullo G., Gullo R., Raspanti C., De Marco P., Porrello C., Profita G., Gulotta G. Deep SSI after mesh-mediated groin hernia repair: management and outcome in an emergency surgery department. G Chir 2017; 38(1): 41–45, https://doi.org/10.11138/gchir/2017.38.1.041.
- Abbaszade T.N., Anisimov A.Yu. Diagnosis and prevention of early postoperative wound complications in patients with large ventral hernias. Meditsinskiy vestnik Bashkortostana 2013; 8(3): 21–25.
- Anisimov A.Yu., Abbaszade T.N. The thermography facilities in the diagnosis of early postoperative vulnerary complications at large ventra hernia repair. Dnevnik kazanskoy meditsinskoy shkoly 2015; 4(10): 5–9.
- Pearce A., Thornton L., Sutton P.A., Walsh C.J. Post-operative C-reactive protein profile following abdominal wall reconstruction with transversus abdominis posterior components separation. Int J Surg Case Rep 2017; 40: 17–19, https://doi.org/10.1016/j.ijscr.2017.08.027.
- Zurnadzhjants V.A., Topchiev M.A., Bondarev V.A., Kchibekov E.A., Serdyukov M.A. Assessment of the course of wound process under the strangulated hernias of the abdominal wall. Kubanskiy nauchnyy meditsinskiy vestnik 2013; 7(142): 132–133.
- Magomedov M.M., Magomedbekov R.E., Ismailov G.M. Systemic inflammatory response in alloplastic treatment of the inguinal hernias. Vestnik novyh medicinskih tehnologij. Elektronnoe izdanie 2017; 2: 139–144.
- Smarzh T.M., Kuznetsov A.V., Ryzhikova S.L., Druzhinina Yu.G., Autenshlyus A.I. Dynamics of cytokines of blood serum and wound discharge in the process of postoperative wound healing depending on the adaptive capabilities of the body. Elektronnyy nauchnyy zhurnal 2016; 9(12): 60–70.
- Smarzh T.M., Kuznetsov A.V., Shestakov V.V., Fedin E.N. The character of early postoperative complications and cytokine dynamics depending on the adaptive capabilities of the body. Al’manakh mirovoy nauki 2017; 5(20): 25–33.
- Levy S., Moszkowicz D., Poghosyan T., Beauchet A., Chandeze M., Vychnevskaia K., Peschaud F., Bouillot J. Comparison of complete versus partial mesh removal for the treatment of chronic mesh infection after abdominal wall hernia repair. Hernia 2018; 22(5): 773–779, https://doi.org/10.1007/s10029-018-1785-1.
- Antonyuk S.M., Achrameev V.B., Andrienko I.B., Lucenko Yu.G., Grintsov G.A. Peculiarities of prophylaxis of postoperative complications in the alloplasty of large and giant postoperative ventral hernias. Universitetskaya klinika 2016; 12(2): 23–26.
- Izmailov A.G., Dobrokvashin S.V., Volkov D.E., Lukoyanychev Е.Е., Pyrkov V.A., Zakirov R.F., Davlet-Kildeev Sh.A. Device for punctures of cavity structures in postoperative period in patients with ventral hernias. Prakticheskaya meditsina 2016; 5(97): 73–76.
- Izmailov A.G., Dobrokvashin S.V., Volkov D.E., Pyrkov V.A., Zakirov R.F., Davlet-Kildeev S.A., Akhmetzyanov R.F. Concept of prevention and treatment of postoperative wound complications in surgical patients. Prakticheskaya meditsina 2017; 6(107): 50–54.
- Shubinets V., Carney M.J., Colen D.L., Mirzabeigi M.N., Weissler J.M., Lanni M.A., Braslow B.M., Fischer J.P., Kovach S.J. Management of infected mesh after abdominal hernia repair: systematic review and single-institution experience. Ann Plast Surg 2018; 80(2): 145–153, https://doi.org/10.1097/sap.0000000000001189.
- Sharma R., Fadaee N., Zarrinkhoo E., Towfigh S. Why we remove mesh. Hernia 2018; 22(6): 953–959, https://doi.org/10.1007/s10029-018-1839-4.
- Kalish Yu.I., Ametov L.Z., Shayusupov A.R., Yigitaliev S.Kh., Kabulov M.K. Chronic paraprosthesis infection after allohernioplasty. Medichnі perspektivi 2016; 21(4): 62–66.
- Chen T., Zhang Y.H., Wang H.L., Chen W., Wang J. Late-onset deep mesh infection: a study of eight cases detected from 2666 consecutive patients with abdominal wall hernia repairs. Chin Med J (Engl) 2016; 129(15): 1870–1872, https://doi.org/10.4103/0366-6999.186651
- Hanna M., Dissanaike S. Mesh ingrowth with concomitant bacterial infection resulting in inability to explant: a failure of mesh salvage. Hernia 2015; 19(2): 339–344, https://doi.org/10.1007/s10029-014-1330-9.
- Panteleev V.S., Zavarukhin V.A., Pogorelova M.P. The use of ultrasound when removing infected mesh implants after hernioplasty. Meditsinskiy vestnik Bashkortostana 2018; 2(74): 24–28.
- Panteleev V.S., Zavarukhin V.A., Pogorelova M.P. Ultrasonic excision of infected mesh implants after hernioplasty. Vestnik eksperimental’noy i klinicheskoy khirurgii 2018; 11(2): 101–104.
- Panteleyev V.S., Zavarukhin V.A., Pogorelova M.P. Ultrasonic excision of infected mesh implants after hernioplasty (clinical case). Kreativnaya khirurgiya i onkologiya 2017; 7(4): 68–72, https://doi.org/10.24060/2076-3093-2017-7-4-68-72.
- Kugler N.W., Bobbs M., Webb T., Carver T.W., Milia D., Paul J.S. A dual-stage approach to contaminated, high-risk ventral hernia repairs. J Surg Res 2016; 204(1): 200–204, https://doi.org/10.1016/j.jss.2016.04.065.
- Bueno-Lledó J., Torregrosa-Gallud A., Sala-Hernandez A., Carbonell-Tatay F., Pastor P.G., Diana S.B., Hernández J.I. Predictors of mesh infection and explantation after abdominal wall hernia repair. Am J Surg 2017; 213(1): 50–57, https://doi.org/10.1016/j.amjsurg.2016.03.007.
- El-Gazzaz G.H., Farag S.H., El-Sayd M.A., Mohamed H.H. The use of synthetic mesh in patients undergoing ventral hernia repair during colorectal resection: risk of infection and recurrence. Asian J Surg 2012; 35(4): 149–153, https://doi.org/10.1016/j.asjsur.2012.06.003.
- McNichols C.H.L., Diaconu S., Liang Y., Ikheloa E., Kumar S., Kumar S., Nam A., Rasko Y. Outcomes of ventral hernia repair with concomitant panniculectomy. Ann Plast Surg 2018; 80(4): 391–394, https://doi.org/10.1097/sap.0000000000001277.
- Nobaek S., Rogmark P., Petersson U. Negative pressure wound therapy for treatment of mesh infection after abdominal surgery: long-term results and patient-reported outcome. Scand J Surg 2017; 106(4): 285–293, https://doi.org/10.1177/1457496917690966.
- Berrevoet F., Vanlander A., Sainz-Barriga M., Rogiers X., Troisi R. Infected large pore meshes may be salvaged by topical negative pressure therapy. Hernia 2013; 17(1): 67–73, https://doi.org/10.1007/s10029-012-0969-3.
- Jakob M.O., Schwarz C., Haltmeier, T., Zindel J., Pinworasarn T., Candinas D., Starlinger P., Beldi G. Mesh-augmented versus direct abdominal closure in patients undergoing open abdomen treatment. Hernia 2018; 22(5): 785–792, https://doi.org/10.1007/s10029-018-1798-9.
- Chatzoulis G., Chatzoulis K., Spyridopoulos P., Pappas P., Ploumis A. Salvage of an infected titanium mesh in a large incisional ventral hernia using medicinal honey and vacuum-assisted closure: a case report and literature review. Hernia 2012; 16(4): 475–479, https://doi.org/10.1007/s10029-010-0767-8.
- Bury K., Śmietański M., Justyna B., Gumiela P., Śmietańska A.I., Owczuk R., Naumiuk L., Samet A., Paradziej-Lukowicz J. Effects of macroporous monofilament mesh on infection in a contaminated field. Langenbecks Arch Surg 2014; 399(7): 873–877, https://doi.org/10.1007/s00423-014-1225-3.
- Lysenko R.B. Prevention and treatment of wound complications at alloplasty of the complex abdominal wall defects. Zaporozhskij medicinskij zhurnal 2016; 3: 71–75, https://doi.org/10.14739/2310-1210.2016.3.76988.
- Al Daoud F., Thayer A., Sachwani Daswani G., Maraqa T., Perinjelil V., Mercer L. Jr. Management of chronic abdominal wall seroma with Doxycycline sclerotherapy using a Negative Pressure Wound Therapy System KCI-V.A.C.Ulta™ — a case report. Int J Surg Case Rep 2018; 51: 25–28, https://doi.org/10.1016/j.ijscr.2018.08.014.
- Khan S.I., Blumrosen G., Vecchio D., Golberg A., McCormack M.C., Yarmush M.L., Hamblin M.R., Austen W.G. Eradication of multidrug-resistant pseudomonas biofilm with pulsed electric fields. Biotechnol Bioeng 2016; 113(3): 643–650, https://doi.org/10.1002/bit.25818.
- Musaev A.I., Isaev E.B., Maksut u. E., Aliev M.J. Infection control in high risk departments. The economic significance of surgical site infection (literature review). Vestnik KGMA imeni I.K. Ahunbaeva 2018; 2: 58–63.
- Natsional’nye klinicheskie rekomendatsii po gerniologii “Pakhovye i posleoperatsionnye gryzhi” [National clinical guidelines on herniology “Inguinal and postoperative hernias”]. Pod red. Ettingera A.P., Shestakova A.L., Gogiya B.Sh. [Ettinger A.P., Shestakov A.L., Gogiya B.Sh. (editors)]. Moscow; 2018.
- Yakovlev S.V., Zhuravleva M.V., Protsenko D.N., Beloborodov V.B., Briko N.I., Brusina E.B., Gusarov V.G., Eliseeva E.V., Zamyatin M.N., Zyryanov S.K., Kukes V.G., Popov D.A., Sidorenko S.V., Suvorova M.P. Antibiotic stewardship program for inpatient care. Clinical guidelines for Moscow hospitals. Consilium Medicum 2017; 19 (7.1. Surgery): 15–51.
- Chattha A., Muste J., Patel A. The impact of hospital volume on clinical and economic outcomes in ventral hernia repair: an analysis with national policy implications. Hernia 2018; 22(5): 793–799, https://doi.org/10.1007/s10029-018-1803-3.
- Cherla D.V., Holihan J.L., Flores-Gonzalez J.R., Lew D.F., Escamilla R.J., Ko T.C., Kao L.S., Liang M.K. Decreasing surgical site infections after ventral hernia repair: a quality-improvement initiative. Surg Infect (Larchmt) 2017; 18(7): 780–786, https://doi.org/10.1089/sur.2017.142.
- Heniford B.T., Ross S.W., Wormer B.A., Walters A.L., Lincourt A.E., Colavita P.D., Kercher K.W., Augenstein V.A. Preperitoneal ventral hernia repair: a decade long prospective observational study with analysis of 1023 patient outcomes. Ann Surg 2018, https://doi.org/10.1097/sla.0000000000002966.
- Saleh S., Plymale M.A., Davenport D.L., Roth J.S. Risk-assessment score and patient optimization as cost predictors for ventral hernia repair. J Am Coll Surg 2018; 226(4): 540-546, https://doi.org/10.1016/j.jamcollsurg.2017.12.022.
- Gaedcke J., Schüler P., Brinker J., Quintel M., Ghadimi M. Emergency repair of giant inguinoscrotal hernia in a septic patient. J Gastrointest Surg 2013; 17(4): 837–839, https://doi.org/10.1007/s11605-012-2136-7.
- Ponomareva Y.V., Volova L.T., Belokonev V.I. The role of tension factor inprosthetic materials biocompatiblity for hernioplasty. Vestnik meditsinskogo instituta “REAVIZ”: reabilitatsiya, vrach i zdorov’e 2016; 4(24): 66–72.
- Pushkin S.Yu., Belokonev V.I., Kovaleva Z.V., Grachev D.B., Gubskiy V.M. The relation between the complication after operations at patients with hernias and ways of hernioplasty and also follow-up diseases. Tol’yattinskiy meditsinskiy konsilium 2016; 3–4: 19–24.
- Belokonev V.I., Vavilov A.V., Guliaev M.G., Grachev D.B. Tactics of treatment of patients with recurrent postoperative incisional hernias. Tavricheskiy mediko-biologicheskiy vestnik 2018; 21(2): 17–23.
- Bratzler D.W., Dellinger E.P., Olsen K.M., Perl T.M., Auwaerter P.G., Bolon M.K., Fish D.N., Napolitano L.M., Sawyer R.G., Slain D., Steinberg J.P., Weinstein R.A. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm 2013; 70(3): 195–283, https://doi.org/10.2146/ajhp120568.
- Poborsky A.N., Drozhzhin E.V., Ponamaryov N.I., Asutayev Sh.J. Pharmacological and economic feasibility of antibiotic therapy for herniatomy complications. Vestnik SurGU. Meditsina 2017; 1(31): 59–61.
- Korableva A.A., Ziganshina L.E. Basic principles of antibacterial prophylaxis in surgery: Cochrane evidence. The Journal of scientific articles “Health and Education Millennium” 2017; 19(7): 138–141, https://doi.org/10.26787/nydha-2226-7425-2017-19-7-138-141.
- Ioannidis O., Paraskevas G., Varnalidis I., Ntoumpara M., Tsigkriki L., Gatzos S., Malakozis S., Papapostolou D., Papadopoulou A., Makrantonakis A., Makrantonakis N. Hernia mesh repair of the anterior abdominal wall and antibiotic chemoprophylaxis: multiple doses of antibiotics failed to prevent or reduce wound infection. Chirurgia (Bucur) 2013; 108(6): 835–839.
- Kokotovic D., Bisgaard T., Helgstrand F. Long-term recurrence and complications associated with elective incisional hernia repair. JAMA 2016; 316(15): 1575–1582, https://doi.org/10.1001/jama.2016.15217.
- Belyansky I., Daes J., Radu V.G., Balasubramanian R., Reza Zahiri H., Weltz A.S., Sibia U.S., Park A., Novitsky Y. A novel approach using the enhanced-view totally extraperitoneal (eTEP) technique for laparoscopic retromuscular hernia repair. Surg Endosc 2018; 32(3): 1525–1532, https://doi.org/10.1007/s00464-017-5840-2.
- Belyansky I., Reza Zahiri H., Sanford Z., Weltz A.S., Park A. Early operative outcomes of endoscopic (eTEP access) robotic-assisted retromuscular abdominal wall hernia repair. Hernia 2018; 22(5): 837–847, https://doi.org/10.1007/s10029-018-1795-z.
- Albino F.P., Patel K.M., Nahabedian M.Y., Sosin M., Attinger C.E., Bhanot P. Does mesh location matter in abdominal wall reconstruction? A systematic review of the literature and a summary of recommendations. Plast Reconstr Surg 2013; 132(5): 1295–1304, https://doi.org/10.1097/prs.0b013e3182a4c393.
- Dietz U.A., Menzel S., Lock J., Wiegering A. The treatment of incisional hernia. Dtsch Arztebl Int 2018; 115(3): 31–37, https://doi.org/10.3238/arztebl.2018.0031.
- Kukosh M.V., Vlasov A.V., Gomozov G.I. Prevention of early incisional complications in ventral hernia prosthetic repair. Novosti khirurgii 2012; 5: 32–37.
- Lembas A.N., Tampey I.I., Kuchinskiy M.V., Baulin A.V., Baulin V.A., Ivanchenko V.V., Velishko L.N., Velishko S.I., Pigovich I.B. Surgical treatment of recurrent incisional ventral hernias. Ukrainskiy zhurnal khirurgii 2013; 4(23): 59–63.
- Krivoschekov E.P., Molchanov M.A., Grigoryev S.G., Romanov V.E., Grigoryeva T.S. Intraoperative prophylactics of sustaining postoperative complications in IPOM-techniques in ventral and umbilical hernias. Aspirantskiy vestnik Povolzh’ya 2017; 1–2: 103–107.
- Naran S., Shakir S., Shestak K.C., Russavage J.M., Nguyen V.T. Components separation for abdominal wall reconstruction in the recalcitrant, high-comorbidity patient: a review of 311 single-surgeon cases. Ann Plast Surg 2018; 80(3): 262–267, https://doi.org/10.1097/sap.0000000000001275.
- Novitsky Y.W., Fayezizadeh M., Majumder A., Neupane R., Elliott H.L., Orenstein S.B. Outcomes of posterior component separation with transversus abdominis muscle release and synthetic mesh sublay reinforcement. Ann Surg 2016; 264(2): 226–232, https://doi.org/10.1097/sla.0000000000001673.
- Mommers E.H., Wegdam J.A., Nienhuijs S.W., de Vries Reilingh T.S. How to perform the endoscopically assisted components separation technique (ECST) for large ventral hernia repair. Hernia 2016; 20(3): 441–447, https://doi.org/10.1007/s10029-016-1485-7.
- Muse T.O., Zwischenberger B.A., Miller M.T., Borman D.A., Davenport D.L., Roth J.S. Outcomes after ventral hernia repair using the rives-stoppa, endoscopic, and open component separation techniques. Am Surg 2018; 84(3): 433–437.
- Switzer N.J., Dykstra M.A., Gill R.S., Lim S., Lester E., de Gara C., Shi X., Birch D.W., Karmali S. Endoscopic versus open component separation: systematic review and meta-analysis. Surg Endosc 2015; 29(4): 787–795, https://doi.org/10.1007/s00464-014-3741-1.
- Cornette B., De Bacquer D., Berrevoet F. Component separation technique for giant incisional hernia: a systematic review. Am J Surg 2018; 215(4): 719–726, https://doi.org/10.1016/j.amjsurg.2017.07.032.
- Scheuerlein H., Thiessen A., Schug-Pass C., Köckerling F. What do we know about component separation techniques for abdominal wall hernia repair? Front Surg 2018; 5: 24, https://doi.org/10.3389/fsurg.2018.00024.
- Malkov I.S., Mukhtarov Z.M., Malkova M.I. Methods of improving treatment results for patients with postoperative ventral hernia. Kazanskij medicinskij zurnal 2014; 95(4): 543–548.
- Mukhtarov Z.M., Malkov I.S., Alishev O.T. Prevention of wound postoperative complications in patients with postoperative ventral hernia. Prakticheskaya meditsina 2014; 5(81): 106–109.
- de Vries F.E.E., Atema J.J., Lapid O., Obdeijn M.C., Boermeester M.A. Closed incision prophylactic negative pressure wound therapy in patients undergoing major complex abdominal wall repair. Hernia 2017; 21(4): 583–589, https://doi.org/10.1007/s10029-017-1620-0.
- Massey L.H., Pathak S., Bhargava A., Smart N.J., Daniels I.R. The use of adjuncts to reduce seroma in open incisional hernia repair: a systematic review. Hernia 2018; 22(2): 273–283, https://doi.org/10.1007/s10029-017-1690-z.
- Gurusamy K.S., Allen V.B. Wound drains after incisional hernia repair. Cochrane Database Syst Rev 2013; 12: CD005570, https://doi.org/10.1002/14651858.cd005570.pub4.
- Krpata D.M., Prabhu A.S., Carbonell A.M., Haskins I.N., Phillips S., Poulose B.K., Rosen M.J. Drain placement does not increase infectious complications after retromuscular ventral hernia repair with synthetic mesh: an AHSQC analysis. J Gastrointest Surg 2017; 21(12): 2083–2089, https://doi.org/10.1007/s11605-017-3601-0.
- Anurov M.V., Titkova S.M., Oettinger A.P. Classification of prostheses for abdominal hernia repair: analytical literature review. Vestnik Rossiyskogo gosudarstvennogo meditsinskogo universiteta 2015; 1: 5–10.
- Kuznetsova M.V., Kuznetsova M.P., Afanasyevskaya E.V., Samartsev V.A. experimental grounds for using collagen-based anti-adhesion barrier coated with biocides for prevention of abdominal surgical infection. Sovremennye tehnologii v medicine 2018; 10(2): 66, https://doi.org/10.17691/stm2018.10.2.07.
- Birindelli A., Sartelli M., Di Saverio S., Coccolini F., Ansaloni L., van Ramshorst G.H., Campanelli G., Khokha V., Moore E.E., Peitzman A., Velmahos G., Moore F.A., Leppaniemi A., Burlew C.C., Biffl W.L., Koike K., Kluger Y., Fraga G.P., Ordonez C.A., Novello M., Agresta F., Sakakushev B., Gerych I., Wani I., Kelly M.D., Gomes C.A., Faro M.P. Jr., Tarasconi A., Demetrashvili Z., Lee J.G., Vettoretto N., Guercioni G., Persiani R., Tranà C., Cui Y., Kok K.Y.Y., Ghnnam W.M., Abbas A.E., Sato N., Marwah S., Rangarajan M., Ben-Ishay O., Adesunkanmi A.R.K., Lohse H.A.S., Kenig J., Mandalà S., Coimbra R., Bhangu A., Suggett N., Biondi A., Portolani N., Baiocchi G., Kirkpatrick A.W., Scibé R., Sugrue M., Chiara O., Catena F. 2017 update of the 1 guidelines for emergency repair of complicated abdominal wall hernias. World J Emerg Surg 2017; 12: 37, https://doi.org/10.1186/s13017-017-0149-y.
- Botezaty A.A., Railianu R.I., Marakutsa E.V., Monul S.G. Functionally reasonable methods of autoplasty in combination with the autodermoplasty in treatment of large and gigantic median hernia. Vestnik Pridnestrovskogo universiteta. Seriya: Mediko-biologicheskie i khimicheskie nauki 2017; 2(56): 12–18.
- Clay L., Stark B., Gunnarsson U., Strigård K. Full-thickness skin graft vs. synthetic mesh in the repair of giant incisional hernia: a randomized controlled multicenter study. Hernia 2018; 22(2): 325–332, https://doi.org/10.1007/s10029-017-1712-x.
- López-Cano M., Quiles M.T., Pereira J.A., Armengol-Carrasco M., Arbós Vía M.A. Complex abdominal wall hernia repair in contaminated surgical fields: factors affecting the choice of prosthesis. Am Surg 2017; 83(6): 583–590.
- Atema J.J., Furnée E.J., Maeda Y., Warusavitarne J., Tanis P.J., Bemelman W.A., Vaizey C.J., Boermeester M.A. Major complex abdominal wall repair in contaminated fields with use of a non-cross-linked biologic mesh: a dual-institutional experience. World J Surg 2017; 41(8): 1993–1999, https://doi.org/10.1007/s00268-017-3962-2.
- Montgomery A., Kallinowski F., Köckerling F. Evidence for replacement of an infected synthetic by a biological mesh in abdominal wall hernia repair. Front Surg 2015; 2: 67, https://doi.org/10.3389/fsurg.2015.00067.
- Primus F.E., Harris H.W. A critical review of biologic mesh use in ventral hernia repairs under contaminated conditions. Hernia 2013; 17(1): 21–30, https://doi.org/10.1007/s10029-012-1037-8.
- Ferzoco S.J. A systematic review of outcomes following repair of complex ventral incisional hernias with biologic mesh. Int Surg 2013; 98(4): 399–408, https://doi.org/10.9738/intsurg-d-12-00002.1.
- Atema J.J., de Vries F.E., Boermeester M.A. Systematic review and meta-analysis of the repair of potentially contaminated and contaminated abdominal wall defects. Am J Surg 2016; 212(5): 982–995, https://doi.org/10.1016/j.amjsurg.2016.05.003.
- Köckerling F., Alam N.N., Antoniou S.A., Daniels I.R., Famiglietti F., Fortelny R.H., Heiss M.M., Kallinowski F., Kyle-Leinhase I., Mayer F., Miserez M., Montgomery A., Morales-Conde S., Muysoms F., Narang S.K., Petter-Puchner A., Reinpold W., Scheuerlein H., Smietanski M., Stechemesser B., Strey C., Woeste G., Smart N.J. What is the evidence for the use of biologic or biosynthetic meshes in abdominal wall reconstruction? Hernia 2018; 22(2): 249–269, https://doi.org/10.1007/s10029-018-1735-y.
- Stoikes N.F.N., Scott J.R., Badhwar A., Deeken C.R., Voeller G.R. Characterization of host response, resorption, and strength properties, and performance in the presence of bacteria for fully absorbable biomaterials for soft tissue repair. Hernia 2017; 21(5): 771–782, https://doi.org/10.1007/s10029-017-1638-3.
- Majumder A., Neupane R., Novitsky Y.W. Antibiotic coating of hernia meshes: the next step toward preventing mesh infection. Surg Technol Int 2015; 27: 147–153.
- Perez-Köhler B., Bayon Y., Bellón J.M. Mesh infection and hernia repair: a review. Surg Infect (Larchmt) 2016; 17(2): 124–137, https://doi.org/10.1089/sur.2015.078.
- Blatnik J.A., Thatiparti T.R., Krpata D.M., Zuckerman S.T., Rosen M.J., von Recum H.A. Infection prevention using affinity polymer-coated, synthetic meshes in a pig hernia model. J Surg Res 2017; 219: 5–10, https://doi.org/10.1016/j.jss.2017.05.003.
- Grafmiller K.T., Zuckerman S.T., Petro C., Liu L., von Recum H.A., Rosen M.J., Korley J.N. Antibiotic-releasing microspheres prevent mesh infection in vivo. J Surg Res 2016; 206(1): 41–47, https://doi.org/10.1016/j.jss.2016.06.099.
- Pérez-Köhler B., García-Moreno F., Brune T., Pascual G., Bellón J.M. Preclinical bioassay of a polypropylene mesh for hernia repair pretreated with antibacterial solutions of chlorhexidine and allicin: an in vivo study. PLoS One 2015; 10(11): e0142768, https://doi.org/10.1371/journal.pone.0142768.
- Ha A., Criman E.T., Kurata W.E., Matsumoto K.W., Pierce L.M. Evaluation of a novel hybrid viable bioprosthetic mesh in a model of mesh infection. Plast Reconstr Surg Glob Open 2017; 5(8): e1418, https://doi.org/10.1097/gox.0000000000001418.
- Mokhov E.M., Kadykov V.A., Sergeev A.N., Askerov E.M., Armasov A.R., Lyubskiy I.V., Serov E.V., Sakharov A.A., Sukhov A.D. Using of a new biologically active thread in a clinical practice. Sovremennye problemy nauki i obrazovaniya 2016; 5: 159. URL: http://science-education.ru/ru/article/view?id=25434.
- Mokhov E.M., Sergeev A.N. Implantation antimicrobial prevention of infection in the surgery intervention area. Sibirskoe meditsinskoe obozrenie 2017; 3(105): 75–81.
- Kornilayev P.G., Plechev V.V. Implantation prevention of pyoinflammatory wound complications in abdominal surgery. Rossiyskiy elektronnyy nauchnyy zhurnal 2013; 1(1): 149–153.
- Plechev V.V., Kornilaev P.G., Feoktistov D.V., Shavaleev R.R., Khakamov T.Sh. Morphological assessment of the efficacy of preventive method for wound complications in implant hernioplasty. Meditsinskiy vestnik Bashkortostana 2014; 9(5): 41–44.
- Parshakov А.А., Gavrilov V.А., Samartsev V.А. Prevention of complications of incisional hernia repair: current problem state (review). Sovremennye tehnologii v medicine 2018; 10(2): 175, https://doi.org/10.17691/stm2018.10.2.21.
- Sadava E.E., Krpata D.M., Gao Y., Novitsky Y.W., Rosen M.J. Does presoaking synthetic mesh in antibiotic solution reduce mesh infections? An experimental study. J Gastrointest Surg 2013; 17(3): 562–568, https://doi.org/10.1007/s11605-012-2099-8.
- Samartsev V.A., Gavrilov V.A., Parshakov A.A., Kuznetsova M.V. Posterior separation hernioplasty TAR in treatment of postoperative ventral hernias W3. Permskiy meditsinskiy zhurnal 2017; 34(1): 35–42.
- Encheva Yu.A., Kuznetsova M.V., Rubtsova E.A., Afanasievskaya E.V., Samartsev V.A. Chlorhexidine and “Prontosan” effect on biofilm formed by Staphylococcus aureus (in vitro study). Permskiy meditsinskiy zhurnal 2015; 32(1): 84–91.










