Genetic Diversity of Autosomal STR Markers in the Brahmin Population of Rajasthan and Haryana: Significance in Population and Forensic Genetics
The aim of the study is to evaluate the suitability of STRs for molecular characterization and forensic applications in unrelated Brahmins of Rajasthan and Haryana states, India.
Materials and Methods. A total of 203 male DNA samples from various districts of Haryana (n=104) and Rajasthan (n=99) were genotyped using the GlobalFiler® PCR Amplification Kit. Allelic frequencies and different forensic parameters like PD, PE, PIC, PM, Ho, He, UHe, and TPI were calculated with different software.
Results. More than 200 alleles were present in both populations, ranging from 6.0 to 35.2 and SE33 was the most polymorphic marker. The combined power of discrimination was 1. To know the relatedness with other Indian Brahmin populations, the UPGMA dendrogram and principal component analysis plot were visualized to show that both populations are close to each other and in nearby Saraswat Brahmins of Himachal Pradesh. This study showed a genetic relationship and forensic examination in the Haryana and Rajasthan Brahmin populations and various ethno-linguistically diverse populations of India.
Conclusion. The results imply that the highly polymorphic 21 autosomal STR loci might be applied for individuals’ forensic identification and parentage testing. This study also suggests that the kit having both autosomal and Y-STR markers is appropriate for a better understanding of the genetic and forensic examination in the Brahmin population of Haryana and Rajasthan.
Introduction
Genetic diversity is essential for the evolution of a species, and it offers the genetic make-up of a species based on the total number of gene distinctiveness features. Diversity reasonably determines population or human social organization in language, ethnicity, culture, geography, social aspect, and communities worldwide. These characteristics are common in the alpine environment, where people frequently live in separate communities far from developed areas [1].
Some traditions emerged due to the assimilation of varied cultures and others died over a period of time. On the other hand, certain cultures have remained steadfast and persistent in demonstrating their presence over time [2]. India has always welcomed people of many racial groups and ethnicities. According to various pieces of evidence, it is possible that modern people left Africa via the southern coastal route and India emerged as a vital human mobility corridor by the initial wave of migration along the south coast route [3]. The interaction and conversion of ideas may have resulted in hybridization between populations of various ethnolinguistic categories in India that further resulted in cultural, linguistic, and genetic variation [4]. In 1919, Ludwik and Hanka Hirszfeld, pioneers in blood typing, first reported the genetic variation among human populations [5]. However, the variations studied in blood groups were insufficient to identify a particular individual from a gene pool. Thus, this area of interest was revolutionized by detecting variations in the human gene pool at the DNA level [6]. In the early days, restriction fragment length polymorphism analysis was employed for DNA analysis. Later on PCR-based assays (with increased sensitivity and power of discrimination) were used along with SNPs, VNTRs, and STRs. In 1990, Alec Jeffrey, with the Human Genome Project, created a wide range of STR sequences [7].
By the mid-1990s, multiple STR markers were added in single multiplexed reactions in forensic DNA testing. Several studies have been used with autosomal STR markers to investigate genetic relationships among Indian groups. The caste system in India is supported by four primary groups or varna: Brahmin, Kshatriya, Vaishya, and Sudra. The castes are further divided into sub-castes and individuals belonging to these sub-castes practice endogamy, a system in which an individual marries inside a specific group [8].
The aim of the study was to investigate the molecular characterization and forensic applications of Brahmins who practice endogamy i.e. they marry within the same caste, practice gotras (clan) system, and do not marry in the same gotras.
Materials and Methods
DNA isolation and ethical consideration. Present study was conducted on stored DNA samples in lab G-22, Department of Genetics, Maharshi Dayanand University (Rohtak, Haryana, India) [9]. A total of 203 male DNA samples from various districts of Haryana (n=104) and Rajasthan (n=99) were used. For autosomal STR analysis in Haryana and Rajasthan Brahmin population, ethical clearance was taken from the Institutional Human Ethical Committee (IHEC) vide letter No.IHEC/2021/289 dated September 9, 2021.
PCR amplification and genotyping. 1.2-mm punch of the FTA card containing the DNA samples was used for amplification [10]. Twenty-one autosomal and three sex-determining markers were amplified in a total volume of 25 μl using the GlobalFiler® PCR Amplification Kit (Thermo Fisher Scientific, USA) according to the manufacturer’s instructions. Throughout the reactions, positive and negative controls were employed. Capillary electrophoresis was used to separate and detect amplified products using an ABI 3500xL Genetic Analyzer (Applied Biosystem, USA). GeneMapper IDX version 1.6 (Applied Biosystem, USA) was utilized for manual allele calling.
Statistical data processing. To calculate the allelic frequency distribution, as well as forensic parameters like polymorphism information content (PIC), probability of match (PM), typical paternity index (TPI), observed heterozygosity (Ho), expected heterozygosity (He), and unbiased heterozygosity (UHe) for 21 autosomal STR loci, STRAF (STR analysis for forensics) online software [11] and GenAlEx 6.5 were used [12]. Additionally, a UPGMA dendrogram [13] was generated based on the Dsw distance determined by POPTREE2 software [14] to examine the genetic distance between the present study population and formerly observed 10 Brahmin groups of India. The principal component analysis (PCA) plot was also created with the help of allelic frequency data through Paleontological Statistics (PAST) software version 3.02 [15] to evaluate the grouping pattern between these populations.
Quality assurance. A positive control DNA template (2800 M), provided along with the kit, and negative controls were used throughout the study. The experimental work was carried out in an accredited laboratory conforming to ISO/IEC 17025 standard. Further, the authors have also qualified International DNA Proficiency Test (http://gitad.ugr.es/principal.htm).
Results and Discussion
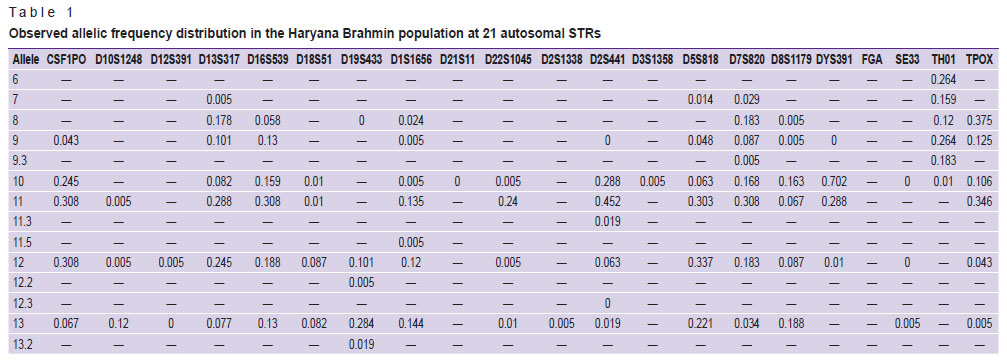
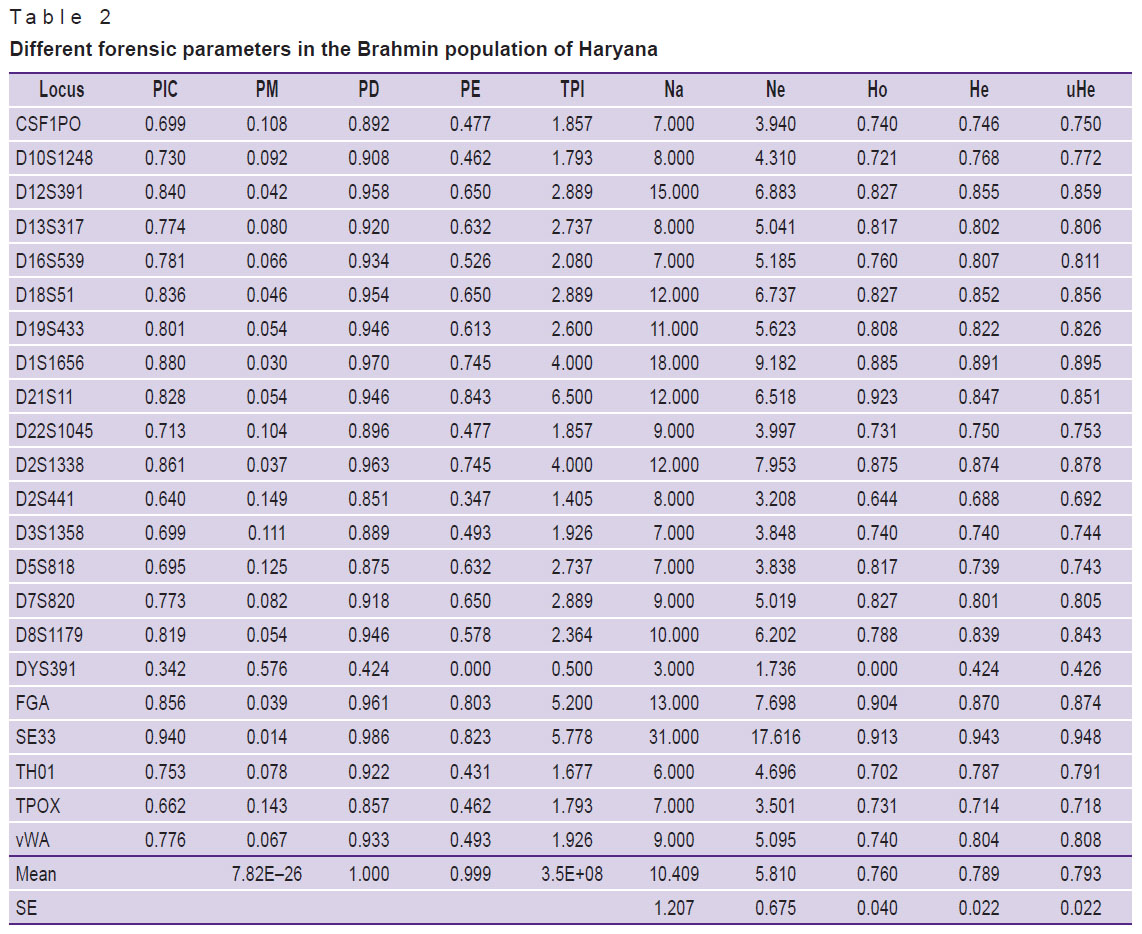
In the Haryana Brahmin population, a total of 61 distinct alleles were detected in the Haryana Brahmin community, ranging from 6.0 to 34.2 and 229 alleles with an average of 10.409. Table 1 shows the allele frequency distribution among Haryana Brahmins. The observed allele frequency ranged from 0.005 to 0.452, showing allele 11 (0.452) as the most common in the study population at the D2S441 locus. The researchers discovered Ho as 0.760±0.040, He as 0.789±0.022, and UHe as 0.793±0.022. Table 2 lists the forensic parameters that were evaluated, including PIC, power of exclusion (PE), power of discrimination (PD), PM and TPI as well as number of alleles (Na), number of effective alleles (Ne), Ho, UHe, and He. With 0.940 (PIC) and 0.986 (PD), locus SE33 was the most polymorphic and discriminative. Moreover, in all 21 autosomal STRs, PD, PE, PIC, PM, Ho, He, and UHe ranged from 0.851 (D2S441) to 0.986 (SE33), 0.347 (D2S441) to 0.843 (D21S11), 0.640 (D2S441) to 0.940 (SE33), 0.030 (D1S1656) to 0.149 (D2S441), 0.644 (D2S441) to 0.923 (D21S11), 0.688 (D2S441) to 0.943 (SE33), and 0.692 (TPOX) to 0.948 (SE33), respectively. The combined power of discrimination and exclusion were 1.0 and 0.823, respectively. As all the samples were confirmed males, 3 out of 104 samples showed the amelogenin Y deletion, i.e. they show only X allele at amelogenin marker. All these samples were also confirmed by the Y-STR profiling.
|
Table 1. Observed allelic frequency distribution in the Haryana Brahmin population at 21 autosomal STRs |
|
Table 2. Different forensic parameters in the Brahmin population of Haryana |
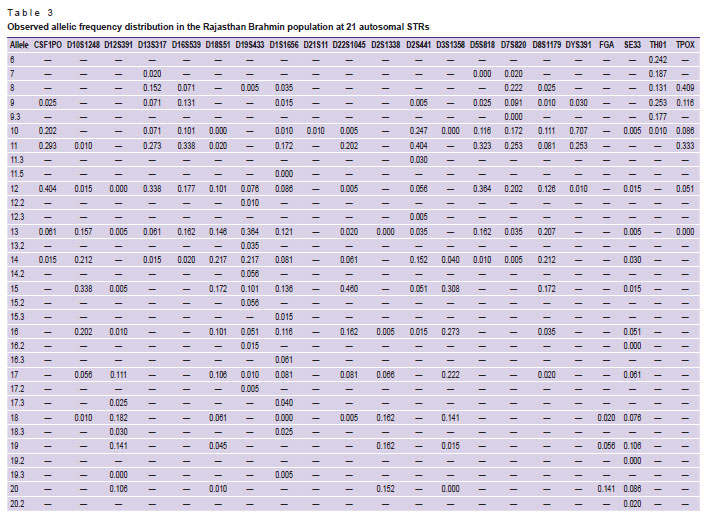
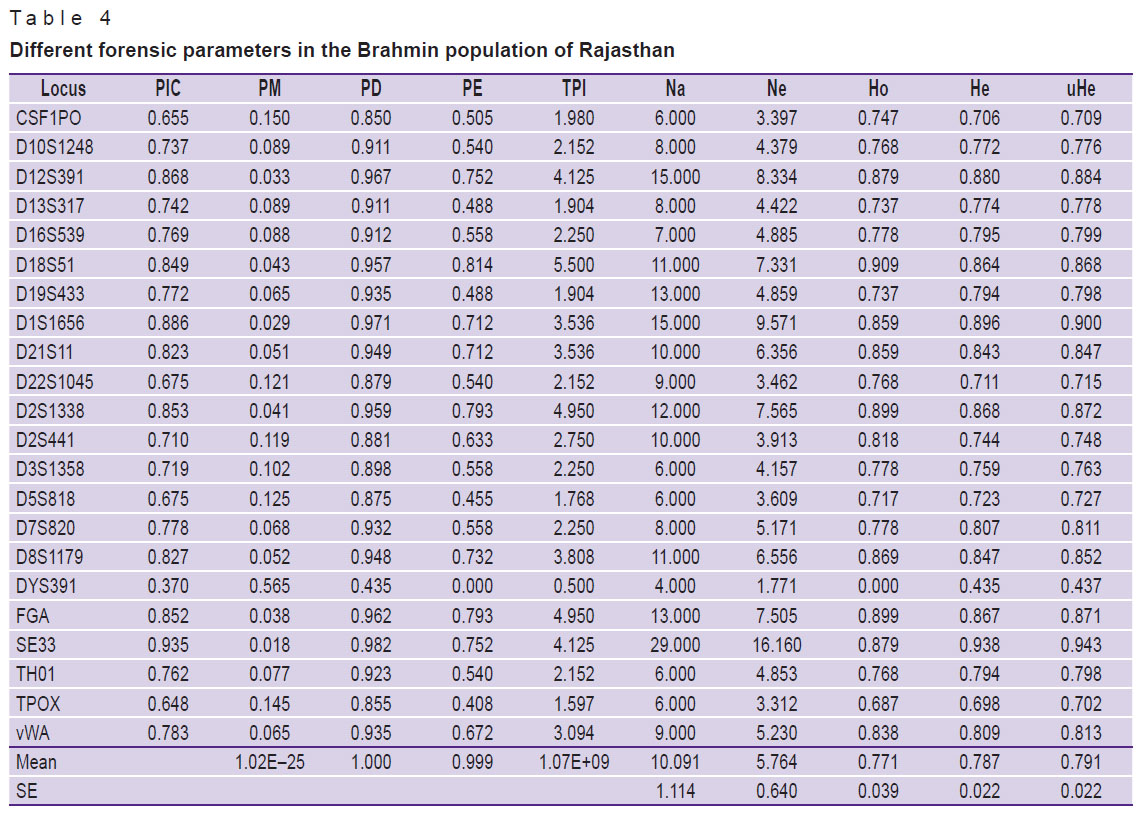
In the Rajasthan Brahmin population, 62 distinct alleles were detected, ranging from 6.0 to 35.2 and a total of 222 alleles with an average of 10.091 alleles per locus were discovered. Table 3 shows the allele frequency distribution among Rajasthan Brahmins. The observed allele frequency ranged from 0.005 to 0.409, showing allele 8 (0.409) as the most common in the examined population and the TPOX locus having the highest allele frequency. It was discovered that the values of Ho as 0.771±0.039, He as 0.787±0.022, and UHe as 0.791±0.022 were present. Table 4 shows the evaluated forensic parameters such as PIC, PD, PE, TPI, and PM, as well as Na, Ne, Ho, He, and UHe. With 0.935 (PIC) and 0.982 (PD), locus SE33 was discovered to be the most polymorphic and discriminative. Moreover, in all 21 autosomal STRs, PD, PE, PIC, PM, Ho, He, and UHe were found ranged from 0.850 (CSF1PO) to 0.982 (SE33), 0.408 (TPOX) to 0.814 (D18S51), 0.648 (TPOX) to 0.935 (SE33), 0.029 (D1S1656) to 0.150 (CSF1PO), 0.687 (TPOX) to 0.909 (D18S51), 0.698 (TPOX) to 0.938 (SE33), and 0.702 (TPOX) to 0.943 (SE33), respectively. The CPD was noted 1 and CPE was 0.999. In this population, only one male out of 100 showed the amelogenin Y deletion [16].
|
Table 3. Observed allelic frequency distribution in the Rajasthan Brahmin population at 21 autosomal STRs |
|
Table 4. Different forensic parameters in the Brahmin population of Rajasthan |
These results imply that 21 autosomal STR loci were more polymorphic to a greater extent and might be utilized for individuals’ forensic identification and parentage testing. The genetic affinity of the examined population, i.e. Brahmin of Rajasthan (RJ) and Brahmin of Haryana (HR), was compared to the previously reported Indian Brahmin population, namely, Saraswat Brahmin of Kashmir (KS), Rajasthan (RJ), Punjab (PB), Jammu (JM) and Himachal Pradesh (HP) [17], Kanyakubj Brahmin of Madhya Pradesh (MP) [18], Brahmin (MP) [19], Desasth Brahmin of Maharashtra (MH), Chitpavan Brahmin (MH) [20], and Iyengar Brahmin of Karnataka (KA) [21] populations, using the UPGMA dendrogram and POPTREE2 software. The UPGMA dendrogram based on Dsw genetic distance revealed that the Brahmin community shared a genetic affinity with Himachal Pradesh’s Saraswat Brahmin (Figure 1).
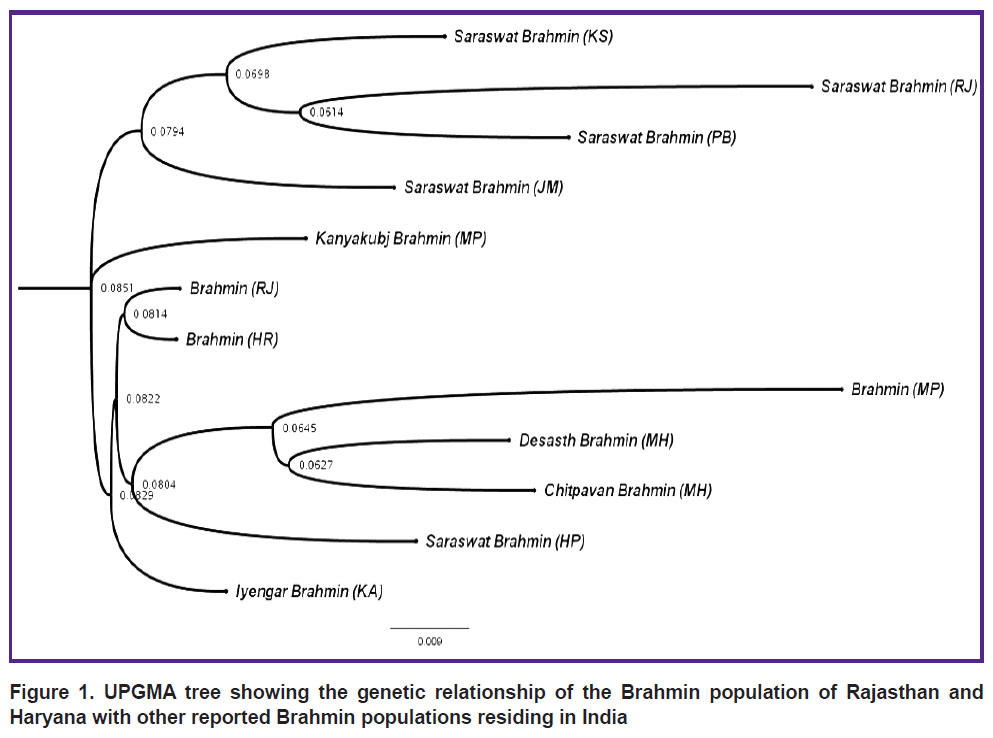
|
Figure 1. UPGMA tree showing the genetic relationship of the Brahmin population of Rajasthan and Haryana with other reported Brahmin populations residing in India |
The PCA result of 10 populations was found to be consistent with the UPGMA dendrogram, indicative of the genetic relatedness of the Brahmin population of Haryana and Rajasthan with other Indian populations (Figure 2).
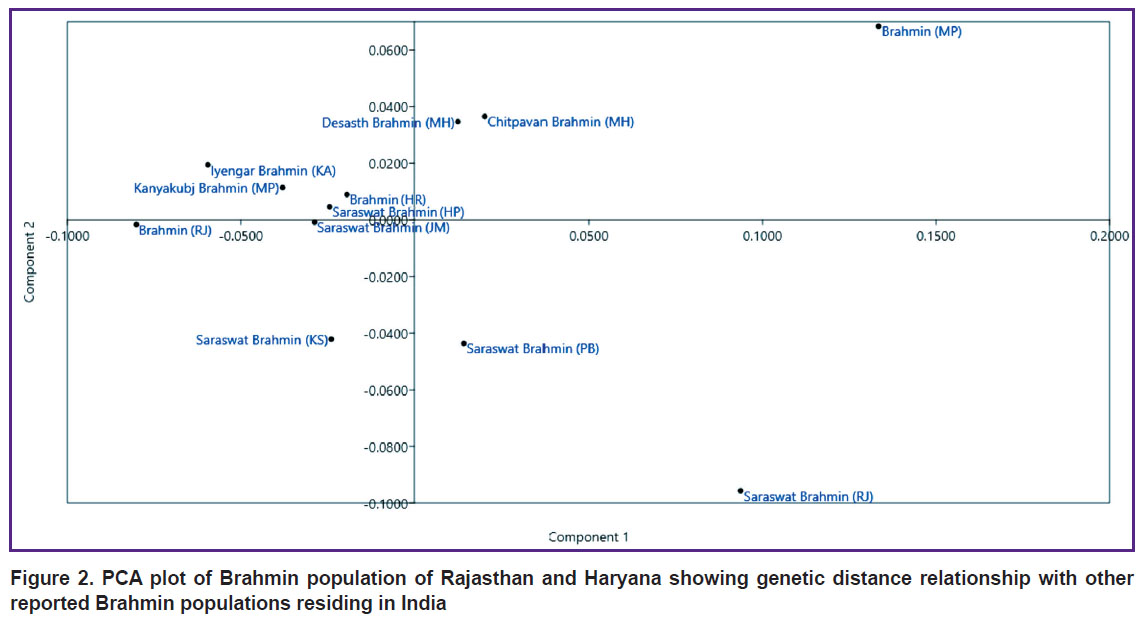
|
Figure 2. PCA plot of Brahmin population of Rajasthan and Haryana showing genetic distance relationship with other reported Brahmin populations residing in India |
Conclusion
This has been a pilot study to bring the genetic characteristics of the Haryana and Rajasthan Brahmin population together and demonstrating the utility of a panel of specific markers in the disputed forensic cases. The genetic data from the present study could be helpful in population genetics and forensics. We suggest further extending this study encompassing a greater more number of samples. It can be conducted by using the representative samples belonging to varied castes/ethnic groups while keeping the immigration routes of the human population in Haryana and Rajasthan states under consideration. In addition, the fine scale understanding of population assignment can be aided by application of genomic data using next generation sequencing. In addition, based on the DNA technology, Regulation bill was enacted by the Ministry of Science and Technology of the Indian Government in Lok Sabha (2019). The presently conducted study would aid in the development of DNA Data Banks at national, as well as state, levels.
Acknowledgement. We thank all sample donors for their contribution to this work. Also, thanks to Thermo Fisher Scientific, India and Mygene Life Solutions LLP, India for providing the kit.
Authors’ contributions. Vivek Sahajpal and Ritu Yadav conceived and designed the experiments. Shivkant Sharma performed all the wet lab experiments. Vivek Sahajpal, Abhishek Singh, and Mukesh Thakur analyzed the data. Ritu Yadav, Shivkant Sharma, and Lokesh Kadian wrote the manuscript. Deepika Bhandari, Shalu Ranga and Chetna Yadav read and improved the manuscript. All the authors participated in the discussion and provided inputs to improve the manuscript’s content. All authors read and approved the final manuscript.
Study funding. The study has not been funded.
Conflicts of interest. The authors have no conflicts of interest to declare that are relevant to the content of this article.
References
- Loewe L., Hill W.G. The population genetics of mutations: good, bad and indifferent. Philos Trans R Soc Lond B Biol Sci 2010; 365(1544): 1153–1167, https://doi.org/10.1098/rstb.2009.0317.
- Singh A., Sahajpal V., Kumar S., Shukla M., Bhandari D., Sharma S., Sharma A., Chandra K., Kumar Sharma L., Thakur M. Genetic evidence of shared ancestry among diverse ethno-linguistic human populations of Himachal Pradesh. Gene 2021; 772: 145373, https://doi.org/10.1016/j.gene.2020.145373.
- Tamang R., Thangaraj K. Genomic view on the peopling of India. Investig Genet 2012; 3(1): 20, https://doi.org/10.1186/2041-2223-3-20.
- Majumdar D.N., Rao C.R. Race elements in Bengal: a quantitative study. Asia Publishing House; 1960.
- Allan T.M. Hirszfeld and the ABO blood groups. Br J Prev Soc Med 1963; 17(4): 166, https://doi.org/10.1136/jech.17.4.166.
- Kandpal V., Saraswathy K.N., Ghosh P.K. Genetic homogeneity among Rajputs of Kangra District, Himachal Pradesh: a study based on surname analysis. South Asian Anthropologist 2011; 11(1): 33–35.
- Gusmão L., Butler J.M., Carracedo A., Gill P., Kayser M., Mayr W.R., Morling N., Prinz M., Roewer L., Tyler-Smith C., Schneider P.M.; DNA Commission of the International Society of Forensic Genetics. DNA Commission of the International Society of Forensic Genetics (ISFG): an update of the recommendations on the use of Y-STRs in forensic analysis. Forensic Sci Int 2006; 157(2–3): 187–197, https://doi.org/10.1016/j.forsciint.2005.04.002.
- Bhatnagar S., Agrawal S. Surname endogamy among the Brahmin of India. Curr Sociol 2002; 50(6): 853–861, https://doi.org/10.1177/0011392102050006005.
- Sharma S., Yadav R., Sahajpal V., Singh M., Ranga S., Kadian L., Yadav C., Patial A., Devi N., Ahuja P. Y-23 mediated genetic data analysis of endogamous Brahmin population of Rajasthan, India. Data in Brief 2022; 42: 108061, https://doi.org/10.1016/j.dib.2022.108061.
- Sahajpal V., Rajput S., Sharma T., Sharma A., Thakar M.K. Development and evaluation of a novel DNA purification buffer and protocol for blood samples on FTA cards. Forensic Sci Int Rep 2019; 1: 100014, https://doi.org/10.1016/j.fsir.2019.100014.
- Gouy A., Zieger M. STRAF — a convenient online tool for STR data evaluation in forensic genetics. Forensic Sci Int Genet 2017; 30: 148–151, https://doi.org/10.1016/j.fsigen.2017.07.007.
- Peakall R., Smouse P.E. GenAlEx 6: genetic analysis in Excel. Population genetic software for teaching and research — an update. Bioinformatics 2012; 28(19): 2537–2539, https://doi.org/10.1093/bioinformatics/bts460.
- Shriver M.D., Jin L., Boerwinkle E., Deka R., Ferrell R.E., Chakraborty R. A novel measure of genetic distance for highly polymorphic tandem repeat loci. Mol Biol Evol 1995; 12(5): 914–920, https://doi.org/10.1093/oxfordjournals.molbev.a040268.
- Takezaki N., Nei M., Tamura K. POPTREE2: Software for constructing population trees from allele frequency data and computing other population statistics with Windows interface. Mol Biol Evol 2010; 27(4): 747–752, https://doi.org/10.1093/molbev/msp312.
- Hammer Ø., Harper D.A.T., Ryan P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol Electron 2001; 4(1): issue1_01.
- Sharma S., Yadav R., Sahajpal V., Kumari L., Sharma A. A comparative study on the frequency of amelogenin Y deletion in a Brahmin population of Haryana and Rajasthan state with other Indian and global populations. Russ J Genet 2022; 58(3): 359–364, https://doi.org/10.1134/s1022795422030139.
- Yadav B., Raina A., Dogra T.D. Genetic proximity of Saraswat Brahmin community of Northern India based on autosomal STR markers. J Bio Innov 2016; 5: 764–785.
- Dubey B., Meganathan P.R., Eaaswarkhanth M., Vasulu T.S., Haque I. Forensic STR profile of two endogamous populations of Madhya Pradesh, India. Leg Med (Tokyo) 2009; 11(1): 41–44, https://doi.org/10.1016/j.legalmed.2008.07.003.
- Gaikwad S., Kashyap V.K. Polymorphism at fifteen hypervariable microsatellite loci in four populations of Maharashtra, India. Forensic Sci Int 2002; 126(3): 267–271, https://doi.org/10.1016/s0379-0738(02)00090-7.
- Ghosh T., Kalpana D., Mukerjee S., Mukherjee M., Sharma A.K., Nath S., Rathod V.R., Thakar M.K., Jha G.N. Genetic diversity of autosomal STRs in eleven populations of India. Forensic Sci Int Genet 2011; 5(3): 259–261, https://doi.org/10.1016/j.fsigen.2010.01.005.
- Rajkumar R., Kashyap V.K. Genetic structure of four socio-culturally diversified caste populations of southwest India and their affinity with related Indian and global groups. BMC Genet 2004; 5(1): 23, https://doi.org/10.1186/1471-2156-5-23.










